Last updated: February 4, 2026
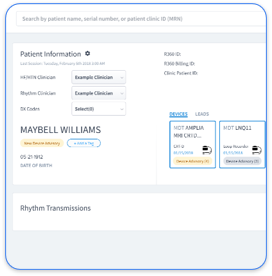
Multi-OEM cardiac device management strains practices that rely on separate OEM portals. Clinicians must log into different systems for Medtronic, Abbott, Boston Scientific, and Biotronik, which creates data silos and extra administrative work. Each manufacturer uses unique transmission protocols and reporting formats, so device technicians juggle multiple interfaces and reconcile inconsistent data views.
Vendor-neutral platforms such as Rhythm360 solve this fragmentation by consolidating all OEM data streams into a single dashboard. Schedule a demo to see how cloud-based integration removes portal switching and standardizes alert workflows. AI-powered computer vision technology normalizes disparate data formats and supports greater than 99.9% transmissibility through redundant feeds and automated gap-filling algorithms.

Interoperability now defines the 2026 device landscape as practices expand their device portfolios. Legacy on-premise systems cannot scale easily or process data in real time for complex multi-vendor environments. Cloud-native platforms provide the performance, security, and flexibility needed to support larger patient panels while preserving high clinical oversight standards.
Device Type | Examples | Key Features | AFib Detection |
CIEDs | Pacemakers, ICDs, ILRs | Continuous monitoring, lead impedance | Episode detection and burden |
Apple Watch | Series 9 | ECG app, PPG monitoring | FDA-cleared irregular rhythm notifications |
Samsung Galaxy | Watch 6 | ECG monitoring, continuous HR | Irregular rhythm alerts |
Fitbit | Versa 4 | 24/7 heart rate tracking | High AFib detection accuracy per Fitbit Heart Study |
Apple Watch Series 5 or 8 used in EQUAL trial improved AFib detection in high-risk older adults through continuous PPG-based monitoring and 30-second ECG capabilities. Fitbit devices achieve high accuracy in detecting AFib episodes per the Fitbit Heart Study, while maintaining FDA clearance for irregular rhythm notifications.
Effective home-based arrhythmia monitoring starts with clear education and simple routines. Managing AFib at home begins with healthy lifestyle changes as a foundational strategy that supports better outcomes and device adherence.
Patient monitoring checklists should include:
Automated messaging systems support adherence with timely reminders and short educational tips. Twilio-integrated platforms allow secure two-way communication while preserving HIPAA compliance and complete audit trails for every patient interaction.
Guideline Category | Recommendation | COR/LOE | 2026 Update |
CIED Monitoring | Remote monitoring for all patients | Class I | Expanded lead management protocols |
AFib Detection | Episodes >6 hours duration | Class I | AI-enhanced burden calculation |
Transmission Frequency | Routine 3-12 month intervals | Class I | Risk-stratified scheduling |
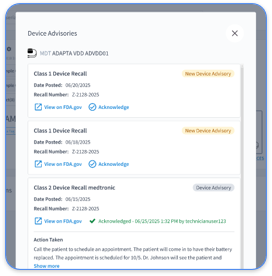
Remote monitoring is a Class I recommendation for all CIED patients according to the 2026 HRS Expert Consensus Statement Update. The guidelines highlight comprehensive lead management and routine transmission schedules that support patient safety and reliable device performance.
Successful RPM programs rely on dedicated teams with clear roles. Practices benefit from certified cardiac device technicians (CCTs) and clinical coordinators who manage daily workflows. HRS Class I recommendations call for structured oversight protocols and defined escalation paths for critical alerts.
Staff training should cover device-specific workflows, CPT code requirements, and emergency response procedures. Teams also need standard operating procedures for documentation, communication, and follow-up timing.
Rhythm360 connects all major OEM portals, including Medtronic, Abbott, Boston Scientific, and Biotronik, through API integration, HL7 messaging, and computer vision technology. The platform supports greater than 99.9% data transmissibility with redundant feeds and automated gap-filling algorithms.
Schedule a demo to see unified multi-vendor workflows that remove portal switching and manual data reconciliation. Teams gain a single source of truth for device data and alerts.
AI-powered processing converts disparate OEM formats into consistent, structured data. Computer vision extracts key values from PDF reports and maps them into standardized fields. Machine learning models flag connectivity problems and potential device malfunctions before they affect patient safety.
Alert management requires the establishment of threshold parameters for each vital sign to distinguish critical alerts needing same-day intervention from routine data. Teams should configure AFib detection thresholds above 6 hours, ventricular tachycardia episode counts, and lead impedance changes that signal clinical concern.
Clear rules reduce low-value notifications and focus staff attention on events that require action. This structure supports faster response times and less burnout.
Bi-directional integration with Epic, Cerner, and Athenahealth keeps documentation accurate and current. HL7 messaging supports real-time synchronization of device data, reports, and notes. These connections maintain audit trails that help with regulatory compliance and quality programs.
Comprehensive education programs increase transmission compliance and more accurate symptom reporting. Automated messaging can deliver medication reminders, lifestyle coaching, and simple troubleshooting tips through HIPAA-compliant channels.
Patients who understand why monitoring matters usually engage more consistently and report issues earlier.
Automated documentation tools capture billable events for CPT codes 93298, 93299, and 99454 while staying aligned with CMS rules. Accurate coding and timely claim submission can raise practice profitability by up to 300%.
CPT Code | Requirements | Rhythm360 Automation |
93298 | Initial setup and patient education | Automated documentation and billing |
93299 | Monthly monitoring and interpretation | AI-generated reports with physician review |
99454 | Device supply and technical support | Inventory tracking and compliance monitoring |
HIPAA-compliant mobile apps let clinicians review transmissions, sign reports, and coordinate care from any location. Optional 24/7/365 oversight by certified cardiac technicians provides continuous monitoring coverage and reduces on-call pressure for in-house staff.
A weekend AFib detection case illustrates this value. Automated alerts flagged a high-burden AFib episode in an asymptomatic patient, which allowed same-day anticoagulation and likely stroke prevention. Without unified monitoring, the event might have remained unnoticed until the next routine review.
Metric | OEM Silos | Rhythm360 |
Data Access | Multiple portals | Single dashboard |
Response Time | High alert fatigue | 80% faster response |
Revenue Impact | Billing losses | 300% revenue boost |
Schedule a Rhythm360 demo to see how vendor-neutral integration reshapes arrhythmia monitoring workflows and removes the operational friction of multi-OEM environments.
Build-versus-buy decisions now favor vendor-neutral platforms for practices that manage several device manufacturers. Rhythm360 implementation usually completes within days to weeks, including EHR integration and staff training. ROI calculations should include lower administrative workload, stronger billing capture, and measurable gains in patient safety.
Implementation readiness checklists should cover EHR compatibility, staff training timelines, patient communication plans, and compliance audit processes. Practices also need clear workflows for alert escalation, device troubleshooting, and emergency coordination before going live.
Remote monitoring devices include CIEDs such as pacemakers, ICDs, and implantable loop recorders, along with consumer wearables like Apple Watch, Samsung Galaxy Watch, and Fitbit devices. These tools provide continuous rhythm surveillance and send physiologic data to clinical teams for timely review and intervention.
Home monitoring of irregular heartbeat involves daily transmission checks, symptom logs, medication adherence tracking, and documentation of lifestyle changes. Patients should keep devices connected, report new or worsening symptoms quickly, and follow prescribed medication plans while participating in structured education programs that build self-management skills.
The 2026 HRS Expert Consensus Statement gives Class I recommendations for remote monitoring of all CIED patients. The document emphasizes comprehensive lead management, routine transmission schedules, and structured clinical oversight. It also defines AFib detection thresholds, transmission frequency expectations, and quality standards for safe device care.
HRS pacemaker follow-up guidelines recommend routine transmissions every 3 to 12 months, depending on device type and patient risk. Remote monitoring supports continuous surveillance between visits and helps teams follow manufacturer guidance and clinical best practices for device longevity and patient safety.
Teams reduce alert fatigue by using AI to customize thresholds, apply clinical significance scores, and automate triage. Effective programs use layered alert levels, role-based routing, and machine learning that separates actionable events from routine data, which improves workflow efficiency.
This 8-step framework for remote arrhythmia monitoring addresses multi-OEM complexity while supporting strong clinical and financial performance. Rhythm360’s vendor-neutral platform removes data silos, cuts alert fatigue, and automates billing compliance to modernize cardiac device management. Schedule a demo to see how unified arrhythmia monitoring can reshape your practice’s approach to remote patient care.