Four levels of interoperability apply directly to cardiac device data management. Foundational interoperability enables basic data exchange between systems without interpretation. Structural interoperability standardizes data format and syntax using standards like FHIR. Semantic interoperability ensures correct data interpretation through standardized vocabularies such as SNOMED CT. Organizational interoperability manages governance, policies, and trust frameworks for secure data exchange across organizations.
In cardiology practices, these levels translate into practical capabilities. Foundational interoperability allows secure transmission of device reports. Structural interoperability enables consistent processing of pacemaker and ICD data. Semantic interoperability ensures accurate interpretation of arrhythmia classifications. Organizational interoperability supports coordinated care across multiple providers and health systems.
The following table shows how each layer of the cardiology technology stack uses standards to address specific interoperability challenges and where Rhythm360 fits.
| Layer | Standards | Challenges | Rhythm360 Solution |
|---|---|---|---|
| Device Data | IEEE 11073 | OEM Fragmentation | AI Normalization (>99.9% via API/HL7/XML/PDF/CV) |
| Transport | HL7 v2/FHIR | Legacy Integration | Bi-directional EHR Integration |
| Workflow | IHE IDCO | Manual Processes | Automated Alert Triage |
| Clinical | SNOMED CT | Alert Fatigue | AI-Powered Prioritization |
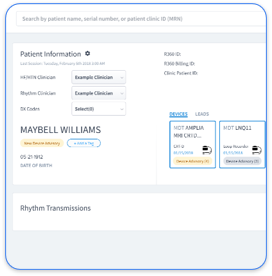
The cardiac device ecosystem still suffers from fundamental interoperability failures. Each OEM maintains proprietary portals that require separate logins, manual data retrieval, and disconnected workflows. Device technicians spend hours in Medtronic CareLink, Boston Scientific LATITUDE, Abbott Merlin.net, and Biotronik Home Monitoring, which creates administrative overhead and reduces time available for patient care.
The global wearable cardiac devices market is projected to reach USD 25.97 billion by 2034, driven by AI integration for arrhythmia detection and real-time monitoring capabilities. However, fragmented interoperability between devices and healthcare platforms reduces data usability for clinical decision-making.
Rhythm360 addresses these challenges as a vendor-neutral bridge that unifies all OEM data streams into a single platform. Key capabilities include AI-powered data reliability, bi-directional EHR integration, mobile accessibility, and automated CPT code capture that drives significant profitability improvements. Unlike competitors such as PaceMate or Implicity, Rhythm360 provides comprehensive coverage across all device types with redundant data feeds that deliver industry-leading transmissibility.

Ready to unify your cardiac data? See how Rhythm360 eliminates OEM silos and automates your workflows.
Healthcare organizations are deepening their use of FHIR for flexible API-based exchange as the standard evolves from R4 to R5. FHIR enables cardiac device data to flow between CIEDs, EHR systems, and clinical decision support tools through standardized resource types such as Observation, Device, and Patient.
The transition from HL7 v2 to FHIR represents a shift from message-based to resource-based data exchange. HL7 v2 requires complex message parsing and custom interfaces. FHIR uses RESTful APIs that support real-time data access and simpler integration. HL7 International's Point-of-Care Device (PoCD) Implementation Guide focuses on cardiac device integration and defines standard methods for transmitting pacemaker, ICD, and implantable loop recorder data.
For cardiology practices, FHIR migration supports automated data ingestion from multiple device manufacturers, real-time alert processing, and seamless EHR integration. The standard's extensibility supports custom cardiac workflows while maintaining interoperability across health systems.
IEEE 11073 provides the semantic foundation for cardiac device interoperability through standardized nomenclature and data models. The standard defines consistent terminology for device parameters, measurements, and alerts across manufacturers, which enables accurate data interpretation regardless of the originating system.
Key components include IEEE 11073-10101 for nomenclature codes that standardize cardiac measurements such as heart rate, arrhythmia classifications, and device status indicators. IEEE 11073-10201 defines the Domain Information Model that structures how cardiac device data is organized and transmitted. These standards ensure that a ventricular tachycardia alert from a Medtronic ICD is interpreted identically to one from a Boston Scientific device.
In daily practice, IEEE 11073 supports automated alert classification, standardized reporting formats, and consistent data mapping across EHR systems. The nomenclature allows AI algorithms to process cardiac data accurately, which supports clinical decision support and predictive analytics.
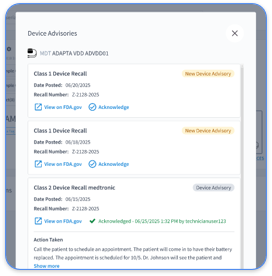
IHE International's Device Technical Framework Revision 10.0, published November 4, 2024, includes the Implantable Device – Cardiac Observation (IDCO) profile as a core specification for device interoperability. The IDCO profile defines standardized workflows for communicating cardiac device observations between systems and supports consistent data exchange in clinical environments.
The IDCO profile (PCD-05) conveys implantable cardiac device observations with context based on IEEE 11073 standards, including nomenclature and Domain Information Model specifications. This approach keeps cardiac device data semantically consistent while supporting diverse clinical workflows.
Real-world applications include automated transmission of device interrogation results, standardized alert formatting, and consistent data presentation across multiple EHR systems. The profile supports both routine monitoring and urgent alert scenarios, which helps practices maintain oversight of their CIED populations while reducing administrative burden.
Successful cardiac device interoperability implementation follows a structured plan that covers technical, clinical, and operational requirements. The process starts with a comprehensive assessment of existing data silos, mapping current workflows, and identifying integration points with EHR systems.
Step-by-step implementation includes evaluating current OEM portal usage and identifying inefficiencies. Teams then map data flows between devices and clinical systems, select FHIR-compliant platforms that support all device manufacturers, configure bi-directional EHR integration with appropriate security controls, test automated billing workflows for the relevant RPM codes, and train staff on unified dashboards and alert management.
The build-versus-buy decision often favors vendor-neutral platforms such as Rhythm360 that provide rapid deployment, broad OEM coverage, and proven ROI. Custom development demands extensive resources, ongoing maintenance, and regulatory compliance management that most practices cannot sustain.
Use the following maturity model to assess your current interoperability level and identify where Rhythm360 can accelerate your progress.
| Level | Description | Self-Evaluation Questions | Rhythm360 Fit |
|---|---|---|---|
| Level 1 | OEM Silos | Multiple portal logins required? | Eliminates all separate logins |
| Level 2 | Basic Integration | Manual data entry into EHR? | Bi-directional automation |
| Level 3 | Workflow Automation | Alert fatigue from noise? | AI-powered triage |
| Level 4 | AI-Enhanced RPM | Advanced data reliability available? | >99.9% data transmissibility |
Common implementation pitfalls include underestimating semantic mapping complexity, performing limited FHIR validation testing, and providing insufficient staff training on new workflows. Healthcare organizations can implement interoperability via a 12–24 month phased roadmap, while vendor-neutral platforms often reduce this timeline to days or weeks.
Scale your RPM program with proven interoperability standards. See Rhythm360's implementation process in action.
Rhythm360 delivers measurable outcomes that reshape cardiology practice operations and financial performance. The platform unifies disparate OEM dashboards into a single interface and dramatically reduces critical alert response times through AI-powered triage that filters non-actionable notifications and prioritizes clinically significant events such as new-onset atrial fibrillation and ventricular tachycardia.
Automated CPT code capture and documentation generation help practices recover previously lost revenue, with implementations reflecting the revenue increases noted earlier. The system maintains compliance with the remote monitoring billing requirements mentioned earlier and preserves comprehensive audit trails for regulatory review.
Hospitals achieve ROI from healthcare interoperability through fewer repeated tests, faster clinical decisions, and reduced administrative load. Rhythm360 extends these benefits to cardiology practices through vendor-neutral data aggregation that removes redundant workflows and supports proactive patient management.
Unlike competitors such as PaceMate that lack comprehensive cardiovascular redundancy, Rhythm360 provides the reliability mentioned earlier through multiple ingestion methods including APIs, HL7 messaging, XML parsing, and computer vision-enabled PDF processing. This reliability helps ensure that critical events are not missed because of technical failures or connectivity issues.
The four levels defined by HIMSS are foundational, structural, semantic, and organizational. Foundational supports basic data exchange. Structural uses standardized formats such as FHIR. Semantic relies on shared meaning through vocabularies like SNOMED CT. Organizational focuses on governance and trust frameworks. In cardiology, these levels support secure device data transmission, consistent processing of pacemaker and ICD information, accurate interpretation of arrhythmia classifications, and coordinated care across multiple providers.
FHIR provides standardized resource types such as Observation, Device, and Patient that allow cardiac device data to move between CIEDs and EHR systems. The standard's RESTful APIs support real-time data access, automated alert processing, and bi-directional integration while preserving semantic consistency across manufacturers and clinical systems.
Standards can solve multi-vendor monitoring when implemented through vendor-neutral platforms. IEEE 11073 supplies standardized nomenclature. FHIR supports consistent data exchange. IHE IDCO defines workflows for cardiac device observations. Successful implementation still requires platforms that normalize data from all OEM sources and present unified clinical interfaces.
The Implantable Device – Cardiac Observation (IDCO) profile defines standardized workflows for communicating cardiac device observations between systems. It supports automated transmission of device interrogation results, standardized alert formatting, and consistent data presentation across EHR systems while maintaining semantic consistency through IEEE 11073 standards.
Vendor-neutral platforms require comprehensive OEM integration, standardized data normalization, and unified clinical interfaces. Key capabilities include API-based data ingestion from all manufacturers, AI-powered alert triage, bi-directional EHR integration, and automated billing compliance. Implementation should prioritize platforms with proven reliability and broad device coverage.
Key trends include FHIR R4/R5 becoming the default standard, AI governance for clinical decision support, unified RPM dashboards with condition-specific pathways, and expanded CMS reimbursement for remote monitoring. The focus shifts from basic compliance to competitive advantage through data-driven care coordination and predictive analytics capabilities.
Cardiac device data interoperability standards now provide a foundation for efficient and profitable cardiology practice operations. FHIR, IEEE 11073, and IHE IDCO enable vendor-neutral data exchange, yet practices still need platforms that unify disparate OEM systems while preserving clinical workflow efficiency.
Rhythm360 connects these standards with real-world implementation, delivering the proven reliability discussed above, the faster alert response discussed earlier, and the substantial revenue gains already outlined through automated workflows and billing compliance. The platform's comprehensive approach to cardiac device interoperability positions practices for sustainable growth in an increasingly complex healthcare landscape.
Ready for scalable, standards-based growth? Transform your cardiac device data management with proven interoperability standards.