CPT 99454 covers device supply, data transmission, collection, and reporting for digitally stored physiological data in remote patient monitoring. The code requires patient use of the RPM device for at least 16 days per 30-day period, with data automatically transmitted from FDA-cleared medical devices.
For cardiology practices managing CIED patients, this includes data from implantable cardioverter defibrillators (ICDs), pacemakers, cardiac resynchronization therapy (CRT) devices, and implantable loop recorders. Heart failure patients using digital scales, blood pressure monitors, and pulse oximeters also generate billable data under CPT 99454 when transmission requirements are met.
The automatic transmission requirement excludes any scenario that relies on manual data entry. Manual patient entry does not qualify for CPT 99454 billing, so vendor-neutral platforms become essential for aggregating OEM data streams from Medtronic, Abbott, Boston Scientific, and Biotronik devices.
CPT 99454 Compliance Checklist:
✓ FDA-approved or FDA-cleared medical device
✓ Automatic data transmission (no manual entry)
✓ Minimum 16 days of data within 30-day billing period
✓ HIPAA-compliant data collection and storage
✓ Single billing per patient per month (regardless of device count)
✓ Documented transmission logs for audit compliance
Common pitfalls include billing CPT 99454 for patients with fewer than 16 transmission days or submitting multiple claims for different devices serving the same patient. Only one CPT 99454 claim is allowed per 30 days per patient, so comprehensive data aggregation becomes crucial for capturing full reimbursement.
The 16-day minimum monitoring duration for CPT 99454 counts any days within a 30-day period when the device transmits physiological data. Measurement days are counted regardless of device type, and different parameters on different days count as separate transmission days.
Consider a heart failure patient who uses a digital scale on 12 days and a blood pressure monitor on 8 days within the same 30-day period. That patient generates 20 total transmission days and qualifies for CPT 99454 billing. CIED patients with daily automatic transmissions usually meet the 16-day threshold, while intermittent monitoring requires closer tracking.
The 2026 introduction of CPT 99445 for 2 to 15 days of data transmission creates a billing option for patients with lower engagement levels. The codes remain mutually exclusive within the same billing period.
Code | Description | Requirements | Billing Frequency |
CPT 99454 | Device supply and data collection | 16+ days transmission | Once per 30 days |
CPT 99457 | Treatment management | ≥20 minutes clinical time | Once per month |
CPT 99454 focuses on device supply and data collection, while CPT 99457 reimburses for the first 20 minutes or more of RPM management time monthly. These codes serve different purposes and can be billed independently for the same patient when documentation clearly supports both services.
CPT 99453 and 99454 can be billed in the same month because they represent distinct services. CPT 99453 covers initial setup and patient education as a one-time fee, while CPT 99454 covers ongoing device supply and data transmission.
Code | Purpose | Frequency | 2026 Reimbursement |
CPT 99453 | Initial setup and education | Once per episode | ~$19 |
CPT 99454 | Device supply and data (16+ days) | Monthly | ~$47 |
CPT 99457 | Treatment management (20+ min) | Monthly | ~$51 |
CPT 99458 | Additional management (20+ min) | Up to 3x monthly | ~$42 |
This combination allows practices to capture both setup costs and ongoing monitoring revenue. The pairing becomes especially valuable when onboarding new CIED patients or expanding RPM services to heart failure populations.
Strong documentation prevents the most common CPT 99454 claim denials and audit findings. Failing to meet the 16-day rule for patient data transmission is one of the most frequent reasons for claim denials, so transmission logs play a central role in compliance.
CPT 99454 Documentation Checklist:
✓ Patient consent for RPM services and data sharing
✓ Device transmission logs showing 16+ days within billing period
✓ FDA device approval documentation or 510(k) clearance
✓ HIPAA-compliant data storage and transmission records
✓ Clinical staff time logs for data review and analysis
✓ Patient communication records for abnormal findings
The 2026 updates keep existing documentation standards in place and add flexibility for telehealth integration. CMS has held reimbursement rates steady for existing RPM codes like 99454, which places more emphasis on documentation quality than on rate changes.
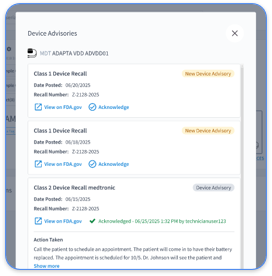
Cardiology practices often struggle with CPT 99454 compliance because of fragmented OEM portals and disconnected device data streams. Multi-vendor environments require staff to log into separate Medtronic CareLink, Abbott Merlin.net, Boston Scientific LATITUDE, and Biotronik Home Monitoring systems, which creates operational inefficiencies and missed transmission tracking.
Rhythm360 solves these challenges with vendor-neutral data aggregation that achieves greater than 99.9% transmissibility through redundant data feeds and AI-powered data normalization. The platform automatically tracks transmission days across all device types, generates compliant documentation, and connects with Epic and Cerner EHR systems to streamline daily workflows.

One electrophysiology clinic using Rhythm360 captured previously lost CPT 99454 revenue by consolidating multi-OEM data into a single dashboard. The clinic reduced administrative overhead by 80 percent and increased RPM billing accuracy. Automated compliance tracking removed manual transmission counting and produced audit-ready documentation for every billable service.
Cardiology teams can automate CPT 99454 compliance and recover lost revenue from fragmented CIED data. Schedule a demo today to see how Rhythm360 reshapes cardiology RPM billing.
Frequent CPT 99454 billing errors include submitting claims for fewer than 16 transmission days, using non-FDA approved devices, and billing multiple claims per patient per month. Billing for manually uploaded device data results in denials, because data must be automatically transmitted from FDA-defined medical devices.
Manual workflows add further compliance risk, especially when staff track transmission days across several OEM portals. Rhythm360’s automated compliance monitoring removes these pitfalls by providing real-time transmission tracking, FDA device verification, and integrated billing documentation that aligns with CMS audit requirements.
The 16-day rule requires at least 16 days of automatic data transmission from FDA-approved devices within a 30-day billing period. Days count whenever any physiological data is transmitted, regardless of device type. For 2 to 15 days of transmission, practices should use the new CPT 99445 code introduced in 2026.
CPT 99454 covers device supply and data collection and requires 16 or more days of transmission. CPT 99457 reimburses for clinical treatment management and requires at least 20 minutes of qualified healthcare professional time. Both codes can be billed independently for the same patient when documentation supports separate services.
CPT 99453 and CPT 99454 can be billed together because they represent separate services. CPT 99453 covers initial setup and patient education and is typically billed once per episode when starting RPM services. CPT 99454 is billed monthly based on meeting transmission requirements.
The 2026 Medicare Physician Fee Schedule keeps CPT 99454 for 16 or more days of data transmission with reimbursement around $47. The major change is the addition of CPT 99445 for 2 to 15 days of transmission, which gives practices billing flexibility for patients with intermittent engagement while maintaining a similar reimbursement rate.
Rhythm360 aggregates data from all major CIED manufacturers into a single platform and tracks transmission days across devices. The system generates compliant documentation, provides real-time compliance monitoring, supports automated billing, and maintains audit-ready records that remove manual tracking errors and increase CPT 99454 revenue capture.
Cardiology practices that master CPT 99454 requirements gain a clear financial and operational advantage. Teams that understand the 16-day transmission rule, maintain strong documentation, and apply the 2026 billing updates can protect revenue while improving patient monitoring. Schedule a demo to see how Rhythm360 turns CPT 99454 compliance into a competitive edge for your cardiology practice.