CPT 99454 represents the monthly supply and data transmission component of remote physiologic monitoring. It covers device-generated data collection for chronic conditions such as heart failure, hypertension, and CIED monitoring. The 2026 CMS Physician Fee Schedule Final Rule clarifies that this non-face-to-face service requires automatic data transmission from FDA-cleared devices and provides approximately $50 in Medicare Part B reimbursement.
CPT Code | Description | Days Required | Reimbursement |
99453 | Initial setup and patient education | One-time only | ~$20 |
99445 | Device supply (2-15 days data) | 2-15 days | ~$50 |
99454 | Device supply (16-30 days data) | 16-30 days | ~$50 |
99457 | Treatment management (20+ minutes) | Monthly | ~$50 |
Billing frequency for CPT 99454 is limited to once per patient every 30 days, regardless of the number of monitoring devices used. The code requires FDA-cleared devices with automatic transmission capabilities and cannot be billed for manually uploaded data.
The 16-day minimum data transmission requirement within each 30-day billing period sits at the core of CPT 99454 compliance. Measurement days are counted regardless of device type, so readings on different days from multiple devices count as separate monitoring days.
Critical Compliance Checklist for the 16-Day Rule:
The 2026 updates make CPT 99445 and 99454 mutually exclusive, so practices cannot bill both codes within the same 30-day period.
Audit-proof CPT 99454 billing depends on complete records that show medical necessity, device performance, and clinical oversight. Documentation standards require detailed tracking of all monitoring activities and related clinical decisions.
Requirement | Description | Cardiology Example | Audit Defense Tip |
Physician Order | Written order documenting medical necessity | Post-CIED implant monitoring for arrhythmia detection | Include specific monitoring parameters and duration. |
Device Setup Logs | Documentation of device shipment and patient training | CardioMEMS sensor activation with patient education | Timestamp all setup activities and patient interactions. |
Transmission Proof | Daily device activity reports showing ≥16 days | Pacemaker interrogation logs with arrhythmia data | Maintain automated transmission receipts and timestamps. |
Clinical Review | Physician sign-off on monitoring data and interventions | EP review of ICD therapy episodes with treatment plan | Document clinical decision-making and patient communication. |
Enhanced documentation requirements for 2026 highlight the need for complete daily device transmission logs and clear medical necessity justification for each billing period.
Insufficient monitoring days, duplicate billing, and non-FDA device usage drive many CPT 99454 denials. Clear internal rules and automated checks help practices avoid these issues and protect revenue.
Top Denial Triggers and Prevention Strategies:
Tracking rejection patterns reveals recurring problems and supports timely appeals with strong clinical documentation.
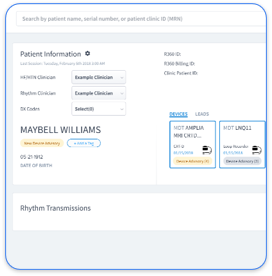
Cardiology practices gain significant value from CPT 99454 through continuous monitoring of cardiac implantable electronic devices and chronic heart failure patients. CIED monitoring includes pacemaker, ICD, and CRT device data transmission. Heart failure programs rely on weight scales, blood pressure monitors, and sensors such as CardioMEMS pulmonary artery monitors.
Cardiology Integration Opportunities:
The vendor-neutral approach removes data silos from manufacturer portals and supports unified care across device brands and monitoring technologies.
Rhythm360’s vendor-neutral platform replaces manual RPM workflows with automated data ingestion, compliance documentation, and accurate billing support for all RPM CPT codes. With RPM markets projected to reach $175 billion by 2030, practices that adopt automation gain a clear operational and financial edge.

The platform delivers more than 99.9% data transmissibility through AI-powered computer vision and redundant data feeds, so critical patient information is not lost due to technical failures. Bi-directional EHR integration with Epic, Cerner, and other major systems removes manual data entry and preserves complete audit trails for compliance documentation.
Revenue Impact Case Study: A multi-physician cardiology practice that implemented Rhythm360 for heart failure monitoring expanded its RPM panel from 50 to 500 patients within six months. The practice achieved a 300% increase in monthly recurring revenue through accurate billing capture and lower administrative overhead.
Schedule a demo to see how Rhythm360’s automated compliance platform can cut billing denials and increase your RPM revenue.
CPT 99454 requires at least 16 days of automatic data transmission within a 30-day billing period. The 2026 CMS updates confirm that measurement days count regardless of device type, so readings on different days from multiple devices count as distinct monitoring days. For monitoring periods with 2 to 15 days of data, practices should bill CPT 99445 instead, since these codes remain mutually exclusive within the same billing period.
CPT 99454 can be billed with evaluation and management (E/M) services when the practice uses the -25 modifier to show a separately identifiable service. The RPM device supply and data transmission covered by 99454 must remain distinct from the E/M service provided during the same encounter. Documentation should clearly separate the ongoing monitoring service from the face-to-face clinical evaluation to support billing both services.
The 2026 CMS updates introduce CPT 99445 for device supply with 2 to 15 days of data transmission, which supports patients with intermittent monitoring compliance. CPT 99470 was added for the first 10 minutes of RPM management time with required patient interaction. These new codes are mutually exclusive with existing codes, so 99445 cannot be billed with 99454 and 99470 cannot be billed with 99457 within the same billing period. The updates keep the 16-day minimum requirement for CPT 99454 while offering alternatives for shorter monitoring periods.
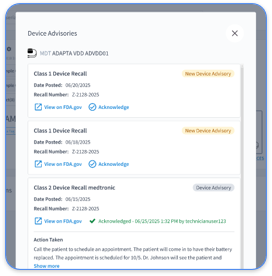
Only FDA-cleared devices with automatic data transmission capabilities qualify for CPT 99454 billing. Eligible devices include cardiac implantable electronic devices such as pacemakers, ICDs, and CRT devices, heart failure monitoring sensors like CardioMEMS, weight scales, blood pressure monitors, and other physiologic monitoring devices that transmit data without manual patient entry. Devices that require manual data upload or manual input do not qualify for RPM billing codes. The devices must collect and transmit physiologic data automatically to the healthcare provider’s monitoring system.
CPT 99454 can be billed once per patient every 30 days, regardless of how many monitoring devices are in use. This monthly billing cycle follows 30-day periods that begin on the initial billing date, not calendar months. Practices cannot submit multiple 99454 claims for the same patient within a single 30-day period, even when several devices track different physiologic parameters. The billing frequency aligns with the requirement for 16 to 30 days of data transmission within each 30-day billing cycle.
Consistent CPT 99454 compliance depends on clear documentation, strict adherence to monitoring duration rules, and careful attention to billing frequency limits. This checklist supports audit-ready remote patient monitoring programs that protect revenue and meet regulatory expectations. Schedule a Rhythm360 demo today to automate compliance and support scalable RPM program growth.