CPT 99454 is a monthly code for RPM device supply and data transmission, and it differs from CPT 99453 and CPT 99457. The code requires a physician order, patient consent, an FDA-cleared device, and 16 unique days of data transmission in a 30-day period. Practices also need an active RPM program, EHR access, and basic billing infrastructure to support consistent use of this code.
CPT 99454 functions differently from related RPM codes that cardiology teams use together. CPT 99453 is billed once for initial device setup and patient education. CPT 99457 requires at least 20 minutes of logged management time each month.
The new 2026 CPT 99445 code covers 2 to 15 days of data transmission and is mutually exclusive with 99454. Cardiology practices that manage CIEDs, pacemakers, ICDs, and CardioMEMS devices can use CPT 99454 to create recurring revenue for heart failure and hypertension monitoring while staying audit-ready.
Proof of 16 Unique Days of Data Transmission
Teams must document proof that monitoring occurred on at least 16 days using an FDA-cleared device. Staff should maintain OEM portal screenshots or automated transmission logs that clearly show each qualifying day.
Step 2: Verify the 16-Day Data Transmission Requirement
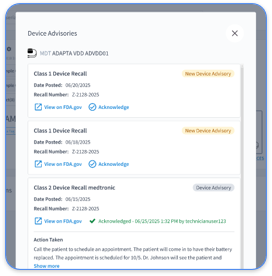
CPT 99454 requires a minimum of 16 days of transmitted physiologic data in a 30-day period for billing. Cardiology practices that manage multiple OEM portals face a heavy administrative burden when they track this manually. Unified dashboards can automatically track transmission compliance across Medtronic, Abbott, and Boston Scientific devices and reduce manual checks.
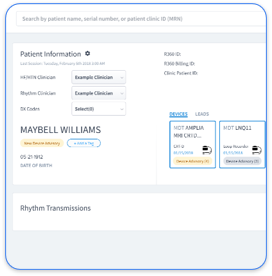
Rhythm360 simplifies multi-vendor RPM billing by automatically ingesting data from all major OEM portals with more than 99.9% transmissibility. The platform generates automated 16-day compliance proofs, consent tracking, and EHR-ready reports. Instead of logging into Medtronic CareLink, Abbott Merlin.net, and Boston Scientific LATITUDE separately, staff see all CIED data in a single dashboard.

Rhythm360 Features That Support CPT 99454 Compliance
Rhythm360 outperforms PaceMate and Implicity by delivering complete cardiac automation instead of partial workflow tools. The platform’s computer vision technology extracts data from unstructured PDFs and provides redundant data feeds when OEM servers experience downtime, which keeps billing and clinical workflows moving.
Schedule a demo to automate your CPT 99454 compliance and see how unified cardiac data management can reduce denials and staff workload.
The year 2026 introduces stricter audit requirements after the CPT 99445 rollout, and auditors will pay closer attention to manual data entry. At the same time, the CMS 2026 MPFS reduces documentation burden by removing some time-based requirements for RPM billing. Practices that rely on automated data capture and clear logs will be better positioned during audits.
Common Denial Reasons and Fixes
Denial Reason | Documentation Fix | Prevention Strategy |
Fewer than 16 days data | Screenshot transmission dashboard | Automated day tracking |
Missing patient consent | Signed consent form with date | Digital consent workflow |
Manual data uploads | Automatic transmission logs | FDA-cleared device verification |
Denial Fix in Practice
Teams should maintain screenshot evidence of transmission dashboards that show the exact days of data receipt. Rhythm360 automatically generates these compliance reports with timestamp verification, which supports fast responses to payer audits.
CPT 99454 requires sixteen unique calendar days of automatically transmitted physiologic data within a 30-day period. Manual data entry, patient-uploaded readings, or missed transmission days do not qualify. The data must come from FDA-cleared medical devices with automatic transmission capabilities, such as implantable cardiac monitors, CardioMEMS sensors, or remote monitoring-enabled pacemakers and ICDs.
Medicare requires FDA-approved devices with automatic data transmission for 16 or more days within a 30-day period. CPT 99454 is mutually exclusive with the new CPT 99445 code, which covers 2 to 15 days of monitoring. Practices must choose one code based on actual transmission days and cannot bill both codes in the same 30-day period. Documentation must prove medical necessity and include complete audit trails that show device type, transmission dates, and clinical use.
Rhythm360 tracks transmission days across all OEM portals and generates compliance reports with clear 16-day verification. The platform pushes structured data directly to EHR systems. It uses AI to fill data gaps, provides backup transmission verification, and creates audit-ready documentation packages. This automation removes manual portal checking and reduces billing errors by up to 99%.
CPT 99454 covers device supply and data transmission and is untimed, based on 16 or more days of data. CPT 99457 covers treatment management and requires at least 20 minutes of logged clinical time each month. CPT 99454 focuses on device functionality and data collection. CPT 99457 involves active clinical review and patient interaction. Practices can bill both codes together when they meet each code’s requirements separately.
Common denials include insufficient OEM portal documentation proving 16 transmission days, missing patient consent forms, billing multiple device codes for the same patient, and using non-FDA-approved devices.
Cardiac-specific denials often stem from fragmented data across multiple OEM portals and difficulty proving transmission compliance for patients with multiple devices. Rhythm360 addresses these issues with unified reporting and automated compliance tracking that consolidates all device data.
Cardiology practices that master CPT 99454 documentation can eliminate many billing denials and grow RPM revenue. Rhythm360’s automated platform supports this goal and has delivered up to 300% revenue uplift for cardiology clients. Schedule a demo to automate your CPT 99454 compliance and transform your cardiac monitoring workflow today.