CPT code 99454 covers device supply with daily recording or alert transmission for remote patient monitoring services. This code represents the monthly supply and data transmission component of RPM billing and remains distinct from initial setup and clinical management services.
Key requirements for CPT 99454 billing include:
Requirement | Details | Cardiology Example |
FDA Device | Auto-transmit physiologic data | Abbott ICD plus HF scale combination |
16+ Days | Data transmission in 30-day period | Medtronic CareLink transmissions |
Monthly Billing | Once per patient per 30 days | Single claim regardless of device count |
These requirements support accurate documentation and consistent compliance for RPM services in cardiology practices.
CPT 99454 requires a minimum of 16 days of transmitted data in a 30-day period for billing, and the 2026 update introduces CPT 99445 for shorter monitoring periods of 2-15 days. These codes remain mutually exclusive based on the actual number of transmission days.
Common cardiology-specific billing pitfalls include:
The 16-day rule remains central for 99454 billing, and CPT 99445 fills the gap for less than 16 days at the same reimbursement rate of approximately $47.
Code | Description | Days/Minutes | Cardiac Use |
99453 | Initial setup and education | One-time | CIED enrollment |
99454 | Device supply | 16+ days | Monthly CIED monitoring |
99445 | Device supply | 2-15 days | Short-term monitoring |
99457 | Clinical review | 20+ minutes | EP consultation |
Clinicians can bill 99453 and 99454 in the same month because these codes represent distinct services. Code 99453 covers initial patient education and setup, and 99454 covers ongoing device supply and data transmission. Each service is billable once per patient per month, regardless of the number of devices used.
Audit-ready documentation for CPT 99454 requires clear records that demonstrate medical necessity and compliance with transmission requirements. Essential documentation includes:
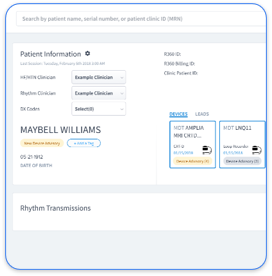
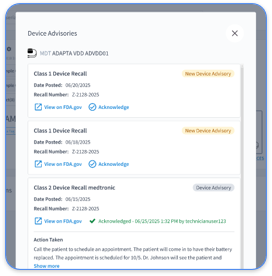
Cardiology practices face unique challenges with device activity logs that must clearly reflect medical necessity for the monitoring period length. Fragmentation across Abbott, Medtronic, and Boston Scientific systems requires unified documentation strategies that reduce audit risk.
Schedule a Rhythm360 demo to see how automated documentation removes manual tracking work while supporting complete audit compliance.
Cardiology practices face complex RPM billing challenges because CIED data management spans multiple OEM platforms. Traditional workflows create data silos where Medtronic CareLink, Abbott Merlin.net, and Boston Scientific LATITUDE systems operate independently, which makes comprehensive patient monitoring and compliant billing difficult.
Rhythm360 closes these gaps through vendor-neutral platform integration that automates 99454 compliance tracking across all major OEM systems. The AI-powered platform achieves greater than 99.9% data transmissibility through redundant data feeds, computer vision parsing, and intelligent gap-filling algorithms.
Key benefits for cardiology practices include:
A representative cardiology practice using Rhythm360 reduced critical alert response times by 80% and achieved flawless 99454 billing compliance across more than 500 CIED patients. The automated platform removed manual portal checking and ensured no billable transmission periods were missed because of data fragmentation.

CMS finalized two new RPM codes for 2026: CPT 99445 for device supply 2-15 days and CPT 99470 for treatment management first 10 minutes. These additions complement existing 99454 billing and address previous gaps in short-duration monitoring.
Key 2026 updates include stricter audit requirements and higher documentation standards for all RPM codes. Approximate reimbursement rates remain around $47 for both 99445 and 99454, which maintains revenue potential while expanding billing opportunities.
Patients | Months | 99454 Reimbursement | Annual Revenue |
100 | 12 | $50 | $60,000 |
250 | 12 | $50 | $150,000 |
500 | 12 | $50 | $300,000 |
Practices that implement Rhythm360's automated compliance platform often see revenue increases of about 300% through stronger billing capture and lower denial rates.
CPT 99454 requires a minimum of 16 days of data transmission within a 30-day period. For monitoring periods with 2-15 days of data, practices should use the new CPT 99445 code introduced in 2026. Both codes have similar reimbursement rates, but remain mutually exclusive based on actual transmission days.
Clinicians can bill CPT 99453 and 99454 in the same month because they represent distinct services. Code 99453 covers initial patient education and device setup, and 99454 covers ongoing device supply and data transmission. Each service addresses a different part of the RPM workflow and carries separate billing requirements.
The national average reimbursement for CPT 99454 in 2026 is approximately $47-60, which aligns with the new CPT 99445 code. Actual rates may vary by geographic location and payer contracts. Consistent reimbursement between codes helps practices maintain revenue regardless of monitoring duration.
CMS requires FDA-cleared devices with automatic data transmission, documented patient consent, and minimum transmission thresholds for billing. Devices must collect physiologic data automatically without manual patient input. Practices must maintain complete documentation of transmission logs and medical necessity for audit compliance.
Bill CPT 99454 once per patient per 30-day period when at least 16 days of data transmission occur. Maintain complete transmission logs, confirm FDA device compliance, and document medical necessity. The code covers device supply and data transmission but excludes clinical management services, which require separate billing codes. Rhythm360's automated platform helps practices meet all guidelines while capturing more RPM revenue.
Ready to simplify 99454 billing and increase revenue for your cardiology program? Schedule a Rhythm360 demo today to see how the automated compliance platform reduces billing errors and supports maximum RPM revenue capture.