Last updated: February 24, 2026
Cardiac device report automation software now sits at the center of life-critical decisions. These platforms handle real-time arrhythmia alerts, device malfunction notifications, and continuous vitals that guide urgent interventions.
Cardiac remote monitoring platforms must process nonstop data streams from multiple OEM portals while staying HIPAA-compliant at every step. The 2026 regulatory landscape adds strict new expectations. Updated HIPAA Security Rules now require full asset inventories, vulnerability scanning every six months, and annual penetration testing.
For cardiac practices, every device manufacturer portal, API connection, and integration point must now meet these security standards. Traditional multi-OEM workflows create unnecessary exposure. Staff log into separate Medtronic CareLink, Abbott Merlin.net, and Boston Scientific LATITUDE portals, and each portal becomes a separate breach risk.
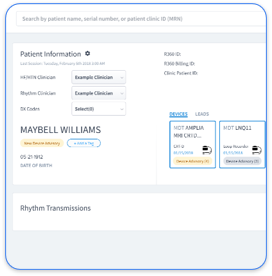
Disparate systems increase alert fatigue and missed critical events. Manual data transcription also raises the chance of human error. Rhythm360 removes these weak points with unified, vendor-neutral data normalization that pulls all CIED data into one secure platform with full audit trails and AI-powered alert triage.
Every cardiac device report automation vendor must sign a detailed Business Associate Agreement that defines ePHI responsibilities and breach notification timelines. Leading RPM platforms maintain SOC 2 certification that aligns with Trust Services Criteria for security, availability, processing integrity, confidentiality, and privacy.
HITRUST certification provides a rigorous framework that covers HIPAA, HITECH, and related regulations. During vendor reviews, request proof of current certifications and annual compliance audits. Rhythm360 operates as a HIPAA-compliant platform that protects patient data across multi-OEM integrations and supports your practice during audits.
The 2026 HIPAA Security Rule requires encryption for all ePHI, including AES-256 at rest, TLS 1.3 in transit, and memory encryption for high-sensitivity workloads. Cardiac device data needs these protections because clinicians rely on it in real time.
Confirm that your software encrypts data from the first device transmission through final EHR storage. Many legacy systems still use TLS 1.2 or weaker methods that no longer meet expectations. Strong key management should include services such as AWS KMS or Azure Key Vault, annual key rotation, and separate keys for each tenant. Rhythm360 uses a secure, HIPAA-compliant infrastructure that protects cardiac device data across the full lifecycle.
Role-Based Access Control limits each user to the patient data required for that role. Clinical staff, device technicians, and administrators see only what they need to perform their work. MFA now applies to all technology assets that access ePHI, not just remote logins.
Configure session timeouts by role. Device technicians may need longer sessions for complex reviews. Administrative users should have shorter timeouts. Enable automatic logoff after inactivity and define emergency access procedures for urgent events. Keep records of access control testing, role assignments, and any changes.
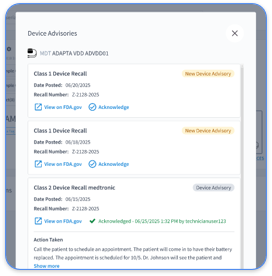
Cardiac device report automation must record detailed audit trails for every access, change, and transmission. New risk analysis expectations include documenting ePHI movement with asset inventories and network maps.
Audit logs should capture user identity, timestamps, specific data viewed, actions taken, and system responses. For cardiac practices, this includes which staff reviewed critical arrhythmia alerts, when reports were generated, and how data moved between OEM portals and EHR systems. Rhythm360 delivers complete audit trails for all ePHI activity so practices can show compliance and maintain clear visibility into access patterns.
Vendor-neutral architecture reduces the risk that comes from juggling multiple OEM portals. Separate Medtronic, Abbott, and Boston Scientific systems each bring different security standards, patch cycles, and breach processes, which increases complexity and exposure.
Vendor-neutral platforms combine these data sources through secure APIs, HL7 messaging, and computer vision that can read unstructured PDF reports. This approach shrinks the attack surface and improves data completeness. Rhythm360 connects with all major CIED manufacturers through encrypted integrations, removes the need for staff to log into OEM portals, and maintains more than 99.9% data transmissibility with redundant feeds and AI-based extrapolation.
Modern cardiac device report automation uses artificial intelligence for both clinical support and security. AI models can flag unusual access behavior, possible data exfiltration, and system anomalies that may signal a breach.
Annual penetration testing now applies to all systems that handle ePHI. Confirm that your vendor performs these tests and shares remediation reports. Rhythm360 uses continuous monitoring, redundant infrastructure, AI-based data reliability checks, and proactive threat mitigation to support uptime and data integrity.
Bi-directional EHR integration removes manual data entry and supports accurate CPT billing. Cardiac device data flows into the EHR, and relevant patient information flows back into the automation platform for full clinical context.
Compliance mapping ensures that automated reports contain all required elements for CPT codes 93298, 93299, and 99454. Integrations must keep audit trails that show data origin and any automated processing. Confirm that EHR connections use encrypted HL7 messaging, strong authentication, and minimum necessary data sharing. Rhythm360 supports these requirements with secure, traceable data exchange.
Rhythm360 delivers a comprehensive, security-first approach to cardiac device report automation. The vendor-neutral platform consolidates data from Medtronic, Abbott, Boston Scientific, and Biotronik through encrypted APIs and computer vision that achieves more than 99.9% data transmissibility.
Compared with competitors such as PaceMate or Implicity, Rhythm360 uses AI-powered triage to cut alert fatigue by up to 80% while maintaining HIPAA compliance. The platform records complete audit trails for every ePHI interaction.
Real-world deployments highlight these advantages. When a patient ICD detects ventricular tachycardia on a weekend, Rhythm360 sends an encrypted push notification through a secure mobile app to the on-call electrophysiologist. The clinician reviews the full arrhythmia episode, checks key history, and coordinates care inside a HIPAA-compliant interface with full logging. This workflow has helped practices reduce critical response times by about 80% and increase revenue by up to 300% through stronger CPT capture. Schedule a demo to see Rhythm360’s security-focused automation in action.

Secure implementation starts with a clear plan and risk review. Begin with an audit of current OEM portal usage, list every system that touches ePHI, and document existing security controls. Capture baseline metrics for alert response times, billing performance, and staff workload.
During vendor selection, request detailed security documentation, current HIPAA compliance details, and BAA templates. Run a pilot with a limited patient group to confirm data accuracy and workflow fit before full rollout. Rhythm360 typically completes implementation, including EHR integration, within days or weeks and provides staff training plus ongoing support.
Risk mitigation should include regular security assessments, staff education on new workflows, and contingency plans for outages or incidents. Document each implementation decision and keep records of security control testing to support future audits.
Rhythm360 operates as a HIPAA-compliant platform and signs Business Associate Agreements with all clients. The platform undergoes ongoing security measures that align with HIPAA Security Rule expectations.
Rhythm360 uses a vendor-neutral security architecture that differs from competitors like PaceMate or Implicity. The platform consolidates data from all major CIED manufacturers through secure integrations and removes the security risks of multiple OEM portals. Rhythm360 provides HIPAA-compliant infrastructure with detailed audit trails.
The 2026 environment requires MFA for all ePHI access, annual asset inventories, vulnerability scanning every six months, and yearly penetration testing. Cardiac device software must also use network segmentation, anti-malware tools, and standardized security configurations. Systems must keep detailed audit logs and support 24-hour breach notification.
Rhythm360 secures each stage of the workflow. The platform tracks all data access and changes with audit trails and uses redundant data feeds for reliability. Vendor-neutral architecture removes the need to log into OEM portals, which reduces attack surfaces while preserving full visibility into patient data through HIPAA-compliant infrastructure.
Healthcare data breaches averaged about $10.3 million in 2025. Cardiac monitoring systems face higher risk because they manage real-time, life-critical data. Third-party vendors account for roughly 80% of healthcare breaches, so vendor choice matters for cardiac practices. The Change Healthcare breach affected about 192.7 million Americans and caused more than $2 billion in financial damage, which shows the scale of potential impact.
This 7-step checklist helps your cardiac practice meet 2026 security requirements while improving efficiency and revenue. HITRUST or SOC 2 validation, AES-256 encryption, vendor-neutral integrations, and complete audit trails work together to protect your patients’ most sensitive data.
Rhythm360 supports these safeguards with a proven, security-first framework. Schedule a demo to see how this HIPAA-compliant, vendor-neutral platform transforms cardiac device report automation while maintaining strong security standards.