Accurate CPT 99454 documentation depends on clear proof of device monitoring and data transmission. This checklist walks your team through each compliance step and gives you language you can adapt directly into your notes.
Step | Requirement | Documentation Template | Proof Example |
1 | Patient Consent | "Patient consented to RPM services on [date]" | Signed consent form in chart |
2 | FDA Device Proof | "Patient using [device model] SN: [number]" | "Omron BP cuff SN: 12345" |
3 | 16+ Days Data Log | "Data transmitted [X] days in 30-day period" | "BP data transmitted 18/31 days" |
4 | Clinical Decision | "Reviewed data, adjusted [medication/plan]" | "Increased lisinopril based on readings" |
5 | Monthly Summary | "Monthly RPM summary completed [date]" | Comprehensive trend analysis |
6 | Audit Trail | "All transmissions logged and reviewed" | Complete transmission history |
Each step needs cardiology-specific documentation that stands up to payer review. For Step 1, patient consent is required at the time of RPM service provision. Example: “Patient John Smith consented to remote blood pressure monitoring on 10/15/26 for hypertension management.”
Step 2 focuses on FDA device verification. Record the exact device model and serial number: “Patient utilizing FDA-approved Omron HeartGuide BP monitor, Serial Number: HG-001234, for daily blood pressure transmission.” This creates a clear, auditable device trail.
Step 3 confirms that you met the 16-day minimum discussed earlier. Document that 16 or more days of data were transmitted in the 30-day billing period. Example: “Patient transmitted blood pressure readings 22 out of 31 days in October 2026, meeting CPT 99454 requirements.”
Steps 4 through 6 show clinical value and oversight. Example language: “Reviewed 22 days of BP readings showing average 145/92 mmHg. Increased lisinopril from 10 mg to 20 mg daily. Provided patient education regarding sodium restriction.” These statements prove active clinical management and support reimbursement.
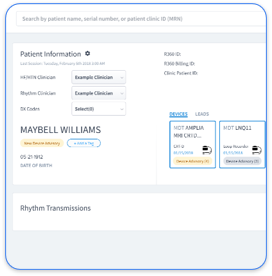
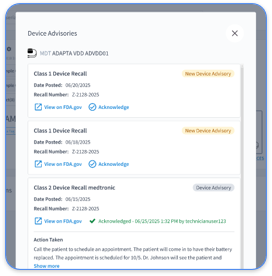
See how Rhythm360 automates all six checklist steps, from device tracking to audit-ready transmission logs.
Manual CPT 99454 documentation adds heavy administrative work and increases audit risk. Rhythm360 reduces that burden through vendor-neutral data ingestion from major device manufacturers including Medtronic, Abbott, Boston Scientific, and Biotronik. The AI-powered system maintains more than 99.9% data transmissibility using redundant feeds and computer vision technology.

Rhythm360’s redundant feeds protect data proofs even when OEM portals experience downtime. The platform creates billing-ready reports, connects with Epic and Cerner EHRs, and produces compliant documentation for CPT 99454 without extra staff effort.
Rhythm360’s cardiology dashboards streamline heart failure and hypertension monitoring by automating the compliance tasks that consume staff time. The platform tracks transmission days and generates monthly summaries, which removes the need for manual log review. It also maintains complete audit trails for every data point so documentation stays audit-ready.
Clinical staff receive prioritized alerts only for actionable events, while routine transmissions are filtered automatically. This targeted alerting reduces alert fatigue by 80% and keeps teams focused on patients who need intervention.
The mobile application lets clinicians review transmissions and sign reports from any location, which supports continuous oversight. Automated patient messaging through the integrated Twilio framework keeps patients engaged and records every communication for compliance.
CPT 99454 billing depends on specific documentation elements that prove CMS compliance and support reimbursement. Medical records must demonstrate monitoring of physiologic metrics with clear clinical justification.
Element | Requirement | Cardiology Note Example |
Device Type | FDA-approved medical device | "FDA-cleared CardioMEMS sensor" |
Data Collection | Automatic electronic upload | "Daily PA pressure readings transmitted" |
Transmission Days | 16+ days in 30-day period | "Transmitted 24/30 days in billing period" |
Clinical Review | Practitioner analysis and action | "Reviewed trends, adjusted diuretic dosing" |
The billing practitioner must personally review transmitted data and record clinical decisions. Generic statements such as “data reviewed” do not meet this standard. Specific actions tied to transmitted data show medical necessity and protect against denials.
CPT 99454 and 99457 serve different roles in remote cardiac care. CPT codes 99457 and 99458 require live, interactive communication with the patient or caregiver, while 99454 covers passive data monitoring.
CPT 99454 focuses on device-generated physiologic data transmission over at least 16 days without live patient interaction. The code includes device supply, setup, patient training, and monthly data analysis. Typical reimbursement averages 58 to 62 dollars per month when documentation is complete.
CPT 99457 requires 20 minutes of live, synchronous two-way audio interaction between practitioner and patient. The code supports treatment management discussions, medication adjustments, and care plan changes. Practices can bill 99457 with 99454 when both services occur in the same period.
Code | Data Days | Live Interaction | Primary Use Case |
99454 | 16+ days | No | Passive device monitoring |
99457 | N/A | Yes (20 min) | Interactive treatment management |
Meeting the 16-day requirement requires systematic documentation of each transmission event. Maintain logs that list transmission dates, times, and key data values. Capture screenshots of device portals or platform dashboards that display transmission calendars as supporting evidence.
Example documentation: “Patient transmitted blood pressure readings on the following dates in October 2026: 10/1, 10/2, 10/4, 10/5, 10/7–10/31 (excluding 10/3, 10/6, 10/15, 10/22, 10/28), totaling 26 transmission days, exceeding the 16-day CPT 99454 requirement.”
Patient consent is required at the time of RPM service provision. Beyond the consent documentation covered in Step 1, the consent form itself should include patient understanding of RPM services, device usage expectations, data transmission frequency, and clinical monitoring protocols.
Consent template: “I consent to remote patient monitoring services including daily blood pressure measurements using an FDA-approved device, automatic data transmission to my healthcare provider, and monthly clinical review of transmitted data for hypertension management. I understand this service will be billed to my insurance using CPT codes 99453 and 99454.”
CMS finalized new RPM codes for 2026 including CPT 99445 for 2–15 days of monitoring, paid at the same rate as 99454. This change gives cardiology programs more flexibility for shorter monitoring periods, which helps with post-discharge cardiac patients.
The 2026 updates also modified setup code 99453 to require at least 2 days of monitoring. This requirement encourages active device use before billing setup services and reduces charges for unused equipment.
The 2026 rules keep RPM and telehealth as distinct services. While CMS streamlined adding services to the Medicare Telehealth Services List, RPM billing requirements remain separate and distinct.
Beyond the new codes, 2026 expanded code stacking opportunities, which now allow RPM codes to be billed with device interrogation codes 93298 and 93299 when both services occur. This expansion creates additional revenue potential for cardiology practices that manage patients with cardiac implantable electronic devices.
Common documentation pitfalls include missing device serial numbers, vague clinical notes, and weak tracking of transmission days. To avoid these errors, record specific device identifiers and clear clinical actions tied to transmitted data, because auditors focus on that level of detail. Generic phrases such as “data reviewed” often fail audits since they do not show the decision-making that supports reimbursement.
Practices using Rhythm360 have reported major gains, including a 300% increase in revenue generation through stronger CPT code capture, better staff efficiency, and easy expansion of RPM service lines for HF and HTN management while avoiding audit penalties. One practice previously managed five device portals manually, which caused missed transmissions and documentation gaps that Rhythm360 later eliminated.
Advanced strategies include stacking CIED device interrogation codes 93298 and 93299 with 99454 and refining heart failure workflows using CardioMEMS sensors. These approaches increase revenue while supporting more comprehensive cardiac care.
CPT 99454 covers remote patient monitoring device supply, setup, and monthly data analysis for patients who meet the 16-day transmission minimum in a 30-day period. The code requires FDA-approved devices, documented patient consent, automatic data upload, and practitioner review with recorded clinical decisions. Billing records must show transmission days, device serial numbers, and actions taken based on the transmitted data.
CPT 99454 covers passive device monitoring without live patient interaction and requires 16 or more days of automatic data transmission. CPT 99457 covers 20 minutes of live, synchronous two-way communication between practitioner and patient for interactive treatment management. Practices can bill both codes together when they provide separate services, with 99454 covering monitoring and 99457 covering live clinical discussion.
Telehealth visits require documentation of live, interactive communication between patient and provider using audio or video technology. Record the platform used, length of the interaction, clinical assessment performed, and treatment decisions made. Include patient location, provider location, and a brief note on connection quality. Telehealth documentation differs from RPM documentation, which centers on device data transmission instead of live interaction.
Yes, CPT 99454 can be billed with telehealth codes when both services occur separately. RPM device monitoring under 99454 and live telehealth consultations serve different purposes and often complement each other. Maintain separate documentation for each service and avoid double-billing for the same work. The RPM portion must still meet the 16-day transmission requirement.
When patients transmit fewer than 16 days of data, use the 2026 CPT 99445 code for 2 to 15 days of monitoring, which pays at the same rate as 99454. If patients transmit data on fewer than 2 days, no RPM codes can be billed. Document reasons for low transmission, such as device malfunction, non-adherence, or connectivity problems, and consider re-education or device replacement.
CPT 99454 does not describe a telemedicine visit with live interaction. It covers passive remote patient monitoring that requires at least 16 days of device data transmission. Using the checklist in this guide helps your team stay compliant with CMS rules and improves reimbursement reliability.
Strong documentation should include patient consent, FDA device verification, transmission day tracking, clear clinical decisions, and a complete audit trail. Rhythm360 automates these requirements through vendor-neutral data integration, AI-driven compliance tracking, and tight EHR integration, which reduces manual errors and uncovers missed revenue in cardiology RPM programs.