Last updated: February 24, 2026
The HIPAA regulatory environment has shifted from 2025 rules, with covered entities required to update their Notice of Privacy Practices by February 16, 2026. These updates focus on substance use disorder records and expanded patient rights disclosures. Cardiology practices face added pressure from CIED data silos across multiple OEM portals, where each manufacturer maintains separate, non-interoperable systems for device monitoring.
Legacy workflows that rely on manual logins to PaceMate, Medtronic CareLink, and Abbott Merlin expose PHI through unsecured processes and weak access controls. Staff often juggle multiple passwords, screenshots, and copy-paste steps, which increases both error rates and breach risk.
The 2026 HIPAA Security Rule changes emphasize enforcement of administrative, physical, and technical safeguards. Practices must run comprehensive gap analyses that compare current protections with new mandates. For RPM workflows, this includes stricter breach notification rules for telehealth PHI and tighter interoperability standards through TEFCA.
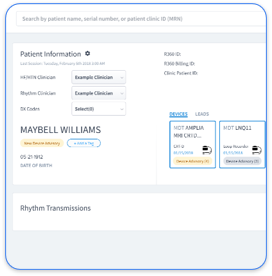
Cardiology teams now manage complex data flows between EHR systems like Epic, device manufacturers, and third-party monitoring services. Each connection must maintain HIPAA compliance at every touchpoint, from device transmission to billing documentation.
This HIPAA compliance checklist outlines 15 essential steps for cardiology practices that manage RPM workflows and CIED data.
1. Appoint HIPAA Privacy and Security Officers
Assign named leaders who own privacy policies, security implementation, and breach response coordination for your practice.
2. Conduct a Comprehensive Risk Assessment
Review all PHI touchpoints, including OEM portals, EHR integrations, mobile devices, and third-party vendors that handle cardiac data.
3. Implement OEM Portal Access Controls
Use role-based access with unique user credentials for each device manufacturer portal and remove shared logins from daily use.
4. Ensure CIED Data Encryption
Confirm encryption in transit and at rest for all cardiac device transmissions, with enterprise-level encryption for device connections and patient engagement.
5. Execute Business Associate Agreements
Secure signed BAAs with every vendor that handles PHI, including device manufacturers, cloud providers, and RPM platforms.
6. Provide Regular Staff Training
Deliver annual HIPAA training with focused modules on RPM workflows, device data handling, and breach prevention scenarios.
7. Develop a Breach Response Plan
Create clear incident response steps for device malfunctions, data transmission failures, and any unauthorized PHI access.
8. Maintain Audit Logs for CPT Billing
Track all RPM activities that support CPT codes 93298, 93299, and 99454 with timestamped access and action records.
9. Secure Mobile Device Access
Require multi-factor authentication and device encryption for smartphones and tablets that access patient data.
10. Establish Data Backup Procedures
Maintain greater than 99.9% uptime with redundant data storage and disaster recovery protocols for critical cardiac monitoring.
11. Update Your Notice of Privacy Practices
Revise your NPP by February 16, 2026, to reflect SUD record protections and expanded patient rights disclosures.
12. Implement Minimum Necessary Standards
Limit PHI access to essential personnel only, using granular permissions for different cardiac monitoring functions.
13. Monitor Third-Party Integrations
Audit EHR connections, API endpoints, and data sharing agreements with external cardiac monitoring services on a regular schedule.
14. Document Patient Consent Processes
Keep clear authorization records for RPM enrollment and any data sharing that extends beyond treatment, payment, and operations.
15. Perform Ongoing Compliance Monitoring
Run quarterly reviews of security measures, access logs, and vendor compliance status, then document findings and remediation steps.
| Safeguard Type | RPM Example | Checklist Action | Risk if Ignored |
|---|---|---|---|
| Administrative | Staff access to device portals | Role-based permissions | Unauthorized PHI access |
| Physical | Workstation security | Screen locks, secure areas | Visual PHI exposure |
| Technical | CIED data transmission | End-to-end encryption | Data interception |
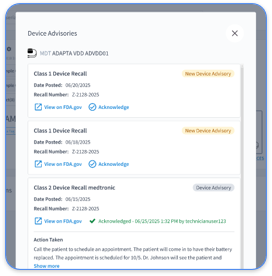
Remote patient monitoring platforms must follow HIPAA, SOC 2, and TEFCA standards with enterprise-level encryption for all device connections and patient engagement workflows. Vendor-neutral RPM solutions that ingest data from multiple CIED manufacturers need strong safeguards such as encrypted APIs, secure data normalization, and AI-powered triage systems that protect PHI during every analysis step.
The 2026 telehealth rules highlight Security Rule enforcement with enhanced administrative, physical, and technical safeguards. Cardiology practices must maintain complete audit trails for all CIED data access and keep encrypted communication channels between devices and servers. Every RPM platform user should authenticate with multi-factor methods.
EHR integration must follow HL7 standards and operate under BAAs that cover bidirectional data flows. These agreements should define responsibilities for encryption, logging, and breach notification.
The most frequent HIPAA violations in cardiology practices include the following issues.
1. Missing OEM Business Associate Agreements
Many practices do not secure complete BAAs with device manufacturers, which creates compliance gaps when PHI passes through manufacturer portals.
2. Manual Data Entry Vulnerabilities
Typing device data manually between systems increases error rates and exposes PHI during copy, paste, and screenshot workflows.
3. Inadequate Mobile Security
Using personal devices or unsecured apps to access cardiac monitoring data introduces risk when encryption and access controls are weak.
4. Insufficient AI Filtering Safeguards
Deploying AI-powered alert systems without PHI sanitization and secure processing environments can create new exposure points.
These violations contribute to a serious trend where 86% of healthcare data breaches in March 2025 were attributed to hacking incidents. OCR penalties now range from tens of thousands to millions of dollars, with 2026 enforcement focusing on poor risk analysis and weak access controls in RPM workflows.
Rhythm360 addresses these compliance challenges with a cloud-based, vendor-neutral platform that maintains greater than 99.9% uptime while applying HIPAA safeguards across all cardiac monitoring workflows. The platform delivers full encryption for data in transit and at rest, automated audit logging for CPT code documentation, and BAA-ready infrastructure that closes compliance gaps between OEM portals and EHR systems.
By consolidating data from all major device manufacturers into a single AI-powered dashboard, Rhythm360 cuts critical alert response times by 80%. Practices also capture up to 300% more revenue through stronger billing documentation and complete audit trails. A recent case study showed how the platform flagged critical atrial fibrillation on a Saturday morning and enabled immediate anticoagulation therapy that prevented a likely stroke. This example illustrates both clinical protection and compliance strength from unified RPM management.

Schedule a demo to see how Rhythm360’s HIPAA-focused architecture turns fragmented cardiac monitoring into streamlined, secure workflows that protect patient data and support practice growth.
Download the comprehensive HIPAA compliance checklist PDF template that includes implementation timelines, vendor evaluation matrices, and audit documentation forms tailored to cardiology RPM workflows. This free resource offers step-by-step guidance for meeting 2026 requirements while improving operational efficiency.
The most significant changes include mandatory Notice of Privacy Practices updates by February 16, 2026, enhanced protections for substance use disorder records, stricter breach notification requirements for telehealth PHI, and expanded enforcement of Security Rule safeguards. Cardiology practices must also follow TEFCA interoperability standards that require minimum-necessary protocols, complete BAAs, and stronger audit logging for electronic health information exchange.
Business associate HIPAA compliance requires signed BAAs with all covered entities and implementation of administrative, physical, and technical safeguards that match covered entity expectations. Vendors must run regular risk assessments across all PHI handling processes, train staff on HIPAA requirements, maintain incident response procedures for breaches, and monitor compliance over time. For RPM vendors, this includes encryption of all cardiac device data, secure API endpoints, detailed audit trails, and clear data retention policies.
The most frequent violations include weak risk assessments that ignore multi-OEM portal vulnerabilities, missing or incomplete BAAs with device manufacturers, and poor access controls that allow unauthorized PHI viewing. Other common gaps involve lack of encryption for CIED data transmission, limited staff training on RPM-specific privacy rules, and weak incident response procedures for device malfunctions or data breaches. These mistakes often lead to OCR penalties that range from thousands to millions of dollars.
Access the comprehensive HIPAA compliance checklist PDF designed for cardiology practices that manage remote patient monitoring workflows. The template includes 2026 regulatory updates, step-by-step implementation guidance, vendor evaluation criteria, audit documentation forms, and compliance tracking tools. This resource helps practices reach HIPAA compliance while improving RPM operations for stronger clinical and financial results.
RPM platforms must implement administrative safeguards such as privacy officers, staff training, and structured access management. They also need physical safeguards that cover workstation security, device controls, and facility access restrictions. Technical safeguards must include encryption, user authentication, audit controls, and secure data transmission.
For cardiac monitoring, platforms must protect encrypted CIED data flows, secure API integrations with EHR systems, and multi-factor authentication for all users. They also need comprehensive audit trails that support CPT code documentation and vendor-neutral data normalization that preserves PHI protection during every analysis step.
HIPAA compliance in 2026 requires more than a basic checklist and demands technology that addresses the specific challenges of cardiac remote monitoring. Rhythm360 delivers a vendor-neutral, AI-powered platform that helps cardiology practices meet compliance requirements while improving clinical outcomes and revenue performance. Schedule your demo today to see how unified RPM management turns compliance into a strategic advantage for your practice.