Last updated: February 24, 2026
HIPAA compliance in healthcare means protecting patient health information through five main rules that govern how covered entities handle protected health information (PHI). The HIPAA Privacy Rule sets federal standards for protecting PHI, including patients' rights to access their information and minimum necessary standards for disclosures. The HIPAA Security Rule requires covered entities to maintain administrative, physical, and technical safeguards that protect the confidentiality, integrity, and availability of electronic PHI. The Breach Notification Rule requires prompt notification to affected patients and HHS after PHI breaches (HHS guidance). The Enforcement Rule establishes penalties up to $1.5 million per violation category annually. The Omnibus Rule extends HIPAA requirements to business associates. For cardiology practices, CIED transmissions, RPM data, and device alerts all qualify as ePHI and require these protections.
This 10-step HIPAA compliance checklist focuses on the realities of cardiology practices that manage RPM and device workflows.
| Step | Requirement | Cardiology Application |
|---|---|---|
| 1 | Conduct comprehensive risk assessment | Evaluate OEM portal vulnerabilities and CIED data flows |
| 2 | Execute business associate agreements | BAAs with device manufacturers and RPM vendors |
| 3 | Implement workforce training | Train staff on PHI handling for device data |
| 4 | Deploy multi-factor authentication | MFA for all OEM portals and monitoring systems |
| 5 | Enable encryption at rest and in transit | Encrypt CIED transmissions and stored alerts |
| 6 | Maintain comprehensive audit logs | Track access to patient device data and reports |
| 7 | Secure RPM data ingestion | Use HIPAA-compliant platforms for device data aggregation |
| 8 | Implement mobile device safeguards | Secure apps for on-call device monitoring |
| 9 | Establish breach response protocols | Define procedures for device data exposure incidents |
| 10 | Perform annual compliance audits | Review OEM access controls and data handling |
Risk assessments must identify areas where PHI could be at risk and assist in implementing effective safeguards. Cardiology teams should revisit these assessments whenever they add new devices, portals, or vendors.
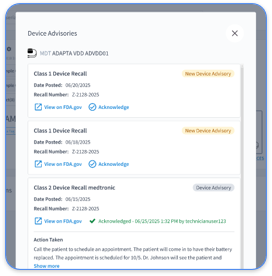
Cardiology and RPM programs often see HIPAA violations tied to device data access, weak encryption, and improper disclosures. 2025 enforcement examples show penalties ranging from $250,000 to $800,000 for violations such as inadequate risk analysis and delayed breach notifications. Cardiology-specific violations include healthcare workers accessing celebrity or family member device records without authorization and storing unencrypted CIED alerts on personal devices.
Other common violations include sharing patient arrhythmia data without proper authorization and using unsecured personal email for device alerts. Staff may also fail to log off OEM portals on shared workstations or discard printed device reports without shredding. Practices sometimes skip business associate agreements with monitoring services or delay breach notifications after device data exposure. Insufficient access controls for device technicians and missing audit trails for critical alert responses also appear frequently. HIPAA penalties range from $157-$1,919 for reasonable cause violations to $78,585-$1,919,173 for willful neglect, with 725 breaches exposing 133 million records in 2023.
The 2025 Security Rule changes propose removing “addressable” implementation specifications and making nearly all safeguards required, including mandatory multi-factor authentication and encryption. New requirements include vulnerability scanning every six months, annual penetration testing, and 24-hour breach reporting by business associates. For RPM and cardiology practices, these changes require encryption of all CIED data transmissions and comprehensive asset inventories that include medical devices.
Practices must also maintain formal incident response plans with annual testing. The reproductive health privacy rule requires signed attestations for certain non-treatment disclosures. Enhanced telehealth enforcement increases scrutiny on RPM billing compliance for CPT codes 99454 and 93298, so documentation and audit trails must stay complete and consistent.
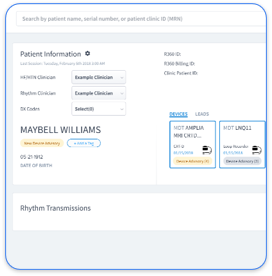
Electronic PHI risks in cardiology often stem from fragmented OEM data silos, unencrypted CIED transmissions, and weak access controls for device monitoring systems. Effective safeguards include end-to-end encryption for all device data and multi-factor authentication for OEM portals. Practices also need comprehensive audit logging of device access and secure mobile applications for on-call monitoring. Schedule a demo to see how Rhythm360 addresses these security challenges with vendor-neutral, HIPAA-compliant monitoring.
| Risk | Example | Required Safeguard |
|---|---|---|
| Data fragmentation | Multiple OEM portals with separate logins | Unified platform with single sign-on |
| Unencrypted transmission | CIED alerts sent via unsecured channels | End-to-end encryption for all data |
| Unauthorized access | Staff accessing non-assigned patient devices | Role-based access controls with audit trails |
Rhythm360 serves as a vendor-neutral, HIPAA-compliant platform built for cardiology practices that manage complex device monitoring workflows. The platform achieves more than 99.9% data transmissibility through AI-powered redundancy and computer vision technology that ingests data from major OEMs including Medtronic, Abbott, Boston Scientific, and Biotronik. Bi-directional EHR integration with Epic and Cerner removes manual data entry and preserves complete audit trails.
AI-powered alert triage cuts critical response times by up to 80% and filters non-actionable notifications to reduce alert fatigue. Automated CPT code capture and documentation for codes 93298, 93299, and 99454 help practices increase revenue by up to 300% through consistent, compliant billing. The secure, HIPAA-compliant mobile application lets clinicians review transmissions and coordinate care from any location while maintaining full audit trails. Unlike competitors PaceMate and Implicity, Rhythm360 offers vendor neutrality without OEM bias.

A recent case study highlights this impact. A Saturday morning atrial fibrillation alert triggered immediate anticoagulation therapy and likely prevented a stroke that could have been missed with fragmented monitoring systems. Schedule a demo to see how Rhythm360 can strengthen your compliance posture while improving outcomes and revenue.
The five main HIPAA rules are the Privacy Rule, Security Rule, Breach Notification Rule, Enforcement Rule, and Omnibus Rule. The Privacy Rule governs PHI use and disclosure, and the Security Rule protects electronic PHI with administrative, physical, and technical safeguards. The Breach Notification Rule requires prompt notification of PHI compromises, and the Enforcement Rule defines penalties for violations. The Omnibus Rule extends requirements to business associates. For cardiology practices, these rules apply to all CIED data, RPM transmissions, and device monitoring activities.
Rhythm360 supports HIPAA compliance through technical, administrative, and physical safeguards that work together. These safeguards include end-to-end encryption, multi-factor authentication, role-based access controls, comprehensive audit logging, secure mobile applications, and business associate agreements with all vendors. The platform also uses regular security assessments and 24/7 monitoring. Its vendor-neutral architecture removes many compliance risks that come from juggling multiple OEM portals.
The 2026 HIPAA updates require multi-factor authentication for all system access and encryption of ePHI at rest and in transit. Organizations must maintain comprehensive asset inventories that include medical devices and perform routine penetration testing. Business associates must report breaches within 24 hours. These changes affect RPM workflows by demanding stronger security for device data transmission and tighter controls on telehealth monitoring platforms.
Small cardiology clinics should start by conducting risk assessments of device monitoring workflows and signing business associate agreements with OEM vendors. Teams need training on PHI handling for CIED data and multi-factor authentication for all portals. Clinics should encrypt device transmissions, maintain audit logs, secure mobile device access, and define breach response procedures. Annual compliance reviews help keep policies current. Unified platforms like Rhythm360 can simplify many of these tasks.
The most common HIPAA violation in cardiology involves unauthorized access to patient device records. Staff may view celebrity, family, or colleague cardiac monitoring data without a legitimate clinical reason. Other frequent violations include using unsecured personal email for device alerts and failing to encrypt CIED transmissions. Practices also see issues with improper disposal of printed device reports and missing audit trails for critical alert responses.
Effective HIPAA compliance healthcare protection starts with this checklist and a prompt risk assessment of your cardiology monitoring workflows. The 2026 regulatory updates require proactive steps, especially for practices that rely on fragmented OEM data streams. Rhythm360's vendor-neutral, HIPAA-compliant platform reduces compliance risk and supports better clinical outcomes and revenue through streamlined workflows and automated billing capture. Schedule a demo today to strengthen your compliance posture and unlock the full potential of unified cardiac monitoring.