The 2026 EHRA and ACNAP clinical consensus statement links structured same-day discharge protocols with ongoing remote pacemaker surveillance, creating a continuous care pathway. This continuum of care requires adherence to five core compliance elements for long-term pacemaker monitoring:
Clinics that align with these guidelines gain predictable compliance through automated tracking in platforms like Rhythm360, which consolidates multi-OEM data streams and maintains regulatory adherence. To see how this consolidation works in your workflow, see how Rhythm360 automates guideline implementation for your practice.
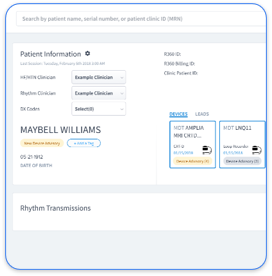
Clinic teams experience heavy administrative burden when they manage separate OEM portals for Medtronic, Abbott, Boston Scientific, and Biotronik devices. Fragmented systems create duplicate data entry, inconsistent documentation, and constant portal switching for cardiac device technicians and administrators.
Alert fatigue grows as notifications arrive from multiple systems without unified prioritization, which delays responses to critical events. Manual documentation across these portals often leads to missed CPT code capture, incomplete audit trails, and measurable revenue leakage.
Routine pacemaker interrogations also become harder to manage as device counts and manufacturers increase. Each additional portal adds complexity to scheduling, tracking, and documenting transmissions across large patient panels.
Rhythm360 addresses these pain points with a single dashboard that unifies data from all supported OEMs and applies AI-powered alert prioritization. This unified view reduces critical response times, cuts manual steps, and supports consistent, guideline-aligned documentation for every device type.

Vendor-neutral platforms differ widely in how they handle data unification, alert management, and compliance automation, so a side-by-side comparison helps clarify tradeoffs. Rhythm360 leads this space by combining multi-OEM data aggregation with AI triage, continuous technician oversight, and deep EHR connectivity.
The platform delivers AI-powered alert triage with 24/7 certified cardiac technician oversight, bi-directional Epic and Cerner integration, and mobile access for clinicians who review transmissions on the go. Clinical practices using Rhythm360 also report substantial revenue gains through more consistent CPT code capture and reduced manual documentation time.
The following comparison highlights how Rhythm360’s comprehensive feature set addresses workflow and compliance gaps that partial solutions leave unresolved:
| Platform | Data Unification (OEMs) | AI Alerts | EHR Integration | Compliance Automation |
|---|---|---|---|---|
| Rhythm360 | All major OEMs (API/HL7/CV, >99.9% transmissibility) | AI triage + CCT oversight (faster response vs. manual workflows) | Bi-directional Epic/Cerner | Automated reports/CPT tracking |
| PaceMate | Partial OEM coverage | Basic filtering | Limited integration | Manual workflows |
| Implicity | Select manufacturers | AI-powered filtering | FHIR connectivity | Automated reporting |
| Murj | Multi-vendor support | Basic notifications | Standard HL7 | Workflow automation |
The single-dashboard approach removes the need for multiple OEM logins and centralizes audit trails, reporting, and billing documentation in one place. To see these differences in a live environment, compare these capabilities in a guided Rhythm360 walkthrough.
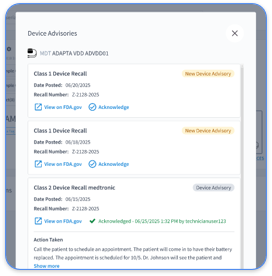
Strong audit readiness depends on consistent documentation of monitoring activities, patient communication, and billing events across the entire care timeline. The IEEE Std 11073-10207-2017/Cor 1-2025 defines standardized structures for medical device data exchange, while CMS evaluation and management guidelines outline what regulators expect to see in documentation for billable services.
The checklist below focuses on four high-impact items that frequently determine audit outcomes and that map directly to automated features in Rhythm360:
| Audit Item | Requirement | Rhythm360 Automation |
|---|---|---|
| Patient ID/Transmission Date | Timestamped logs with device serial numbers | Automated Twilio/AI logging |
| Clinician Review | Physician signature and review date | Mobile e-signature capability |
| Billing Codes | Accurate CPT code capture and justification | Automated code generation |
| Audit Trail | Complete activity history and modifications | Real-time EHR synchronization |
Best practices include maintaining chronological documentation of all patient encounters and ensuring medical necessity justification for each billable service, both of which depend on reliable backup systems for critical data. Because these manual processes consume time and invite errors, automated platforms significantly shorten audit preparation while preserving comprehensive compliance coverage.
Clinics that want to benchmark their current documentation against this framework can request a personalized compliance and audit-readiness assessment.
Successful pacemaker monitoring compliance programs follow a structured three-phase rollout: assessment, EHR integration, and staff training. The assessment phase, which typically takes 2 to 5 days, evaluates current OEM portal usage, identifies workflow bottlenecks, and maps existing documentation and billing processes.
The EHR integration phase, usually 1 to 3 weeks, focuses on configuring bi-directional data flow, setting up APIs, and assigning user roles and access. This phase establishes the technical foundation that supports automated documentation, alert routing, and billing workflows.
Rhythm360’s onboarding process supports each phase with dedicated implementation specialists and pre-configured templates for common clinic workflows. Scalable SaaS deployment adapts to practice size and patient volume, so small clinics and large health systems follow the same structured path.
The platform’s mobile-first design allows clinicians to review alerts and sign reports from any location, which produces immediate productivity gains. Clear training paths, role-based education, and ongoing support then translate these early wins into long-term adoption and sustained compliance performance.
Clinics that master 2026 pacemaker monitoring requirements combine automated workflows, vendor-neutral data consolidation, and disciplined audit preparation into a single, repeatable process. Rhythm360’s platform supports this model by aligning documentation, alert management, and billing with current guidelines while improving response times and revenue capture.
Practices ready to modernize their pacemaker monitoring programs can start their compliance transformation with a tailored Rhythm360 implementation plan.
CMS guidelines define maximum frequencies for routine payment of transtelephonic pacemaker monitoring that vary by device type and time after implant. For example, eligible Guideline II pacemakers may qualify for monitoring every 12 weeks after the first month, as outlined in these guidelines.
These schedules support timely detection of device malfunctions, battery depletion, and arrhythmic events while maintaining Medicare billing compliance. Some patients still require more frequent checks based on clinical indications, device characteristics, or recent programming changes, and urgent transmissions can occur at any time when symptoms or device alerts arise.
Pacemaker interrogation documentation must include physician orders with clear clinical indications, patient consent for remote monitoring, and timestamped transmission logs. Records should also contain comprehensive device parameter reports, clinician review signatures with dates, and appropriate CPT code justification.
Documentation needs to demonstrate medical necessity, reference relevant history and symptoms, and describe any programming changes made during the encounter. All records must remain legible, chronologically organized, and easily accessible for regulatory review or audit.
Effective CIED compliance tools provide vendor-neutral data consolidation, automated documentation workflows, AI-driven alert management, and seamless EHR integration. Rhythm360 offers this combination through multi-OEM data support, high data transmissibility across major manufacturers, automated CPT code capture, and bi-directional Epic and Cerner connectivity.
Key evaluation criteria include mobile access, 24/7 technical support, scalable pricing, and demonstrated ROI from reduced administrative burden and stronger revenue capture.
Audit preparation relies on systematic documentation of monitoring activities, regular internal reviews, and staff training on documentation standards. Clinics should maintain complete patient records with timestamped transmissions, physician signatures, billing justifications, and communication logs for each encounter.
Regular mock audits help uncover documentation gaps before external review. Automated platforms like Rhythm360 support this work with real-time compliance monitoring, structured workflows, and audit-ready reports that align with CMS and industry standards.
Automated pacemaker monitoring platforms typically generate ROI through lower administrative costs, more accurate billing, and improved clinical efficiency. Rhythm360 users report large reductions in time to respond to critical alerts, significant increases in revenue from more consistent CPT code capture, and fewer staff hours spent on manual documentation.
Additional benefits include lower audit risk, faster patient response times, and improved staff satisfaction due to reduced administrative burden. Many practices see positive ROI within 3 to 6 months as workflow efficiency and revenue performance improve.