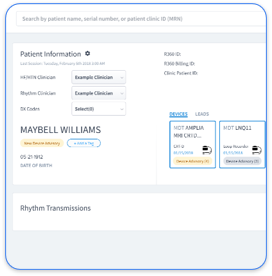
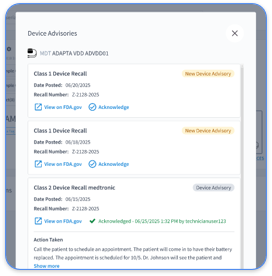
The remote patient monitoring ecosystem has shifted from fragmented OEM portals to AI-powered automation platforms. Manual work across multiple device manufacturer portals creates administrative burden and documentation gaps that often cause claim denials. CMS guidelines allow verbal consent if properly documented, and practices must notify patients about data sharing and opt-out rights to stay compliant.
The 2026 environment favors vendor-neutral platforms that consolidate Medtronic, Abbott, and Boston Scientific data streams while automating consent tracking and documentation. Practices that keep fragmented manual workflows face higher audit scrutiny and revenue loss from undocumented or incomplete consent. Modern platforms such as Rhythm360 solve these problems with automated documentation, EHR integration, and complete audit trails that support consistent 99454 billing compliance.
See how Rhythm360 consolidates your consent workflows
CPT 99454 covers monthly remote patient monitoring device supply and setup with scheduled recordings of physiologic data for at least 16 days in a 30-day period. Cardiology practices use this code for heart failure, hypertension, and cardiac implantable electronic device patients who rely on FDA-approved medical devices that automatically upload data to secure platforms.
The 16-day rule requires documented data transmission for a minimum of 16 days within each 30-day billing period. Practices must confirm that devices qualify as medical devices under FDA regulations and that physiologic data is electronically collected and automatically uploaded for physician review. Common cardiology use cases include weight scales for heart failure monitoring, blood pressure cuffs for hypertension management, and specialized sensors for post-implant device monitoring.
CMS defines specific consent elements that must appear in the record for compliant 99454 billing. Patient consent can be obtained by auxiliary personnel under general supervision, so physicians do not need to be present during the consent conversation.
Essential consent requirements include:
Auxiliary personnel, including employees, independent contractors, or leased employees, can obtain consent under general supervision of the billing practitioner. Now that you understand what CMS requires, the next step is to apply these rules in daily workflows so every consent meets audit standards.
Use this five-step checklist to create clear, consistent consent documentation that stands up to audits.
Use this practical consent template script: “I consent to remote patient monitoring via [specific device] for my [heart failure/hypertension] condition. I understand data will be shared with my healthcare team, potential costs may apply, and I can opt out at any time by contacting the practice.”
Maintain embeddable checklists in your EHR system so every staff member follows the same steps when obtaining consent. This systematic approach reduces audit risk and helps ensure each 99454 claim includes complete consent documentation.
Clear billing rules protect revenue and reduce preventable claim denials. The table below highlights the three most critical rules that determine whether your 99454 claims qualify for payment.
Billing Rule | Details | Cardiology Example |
Frequency | Once per calendar month per patient | Heart failure patient with daily weight monitoring |
Bundling with 99453 | Initial setup can be billed in the same month | First month: 99453 + 99454 |
16-day minimum | Data transmission required for at least 16 days per month | Blood pressure readings on 16 or more days |
Practices can bill 99453 and 99454 together for new RPM patients when both setup and monitoring occur in the same month. CPT 99454 can be billed once per calendar month per patient when the 16-day data transmission requirement is met. New CPT 99445 covers 2 to 15 days of data transmission, and payers treat it as mutually exclusive with 99454.
Rhythm360 turns manual consent tracking into automated compliance through AI-powered documentation and seamless EHR integration with Epic, Cerner, and other major systems. The platform delivers over 99.9% data transmissibility, reduces administrative burden, and supports audit-ready consent documentation for every 99454 claim.

Key features that accelerate compliance:
Case study results show how this works in practice. An electrophysiology clinic eliminated all consent-related claim denials and added comprehensive heart failure RPM services within weeks of implementation. This rapid expansion was possible because Rhythm360’s multi-OEM integrations, covering Medtronic, Abbott, Boston Scientific, and Biotronik in a single platform, removed device compatibility barriers that competitors such as PaceMate and Implicity create through narrower vendor partnerships.
Practices using Rhythm360 report revenue increases of about 300% through stronger CPT code capture, automated billing documentation, and streamlined workflows that free clinical staff to focus on patient care instead of paperwork.
Discover how leading practices achieve audit-proof documentation
Most build versus buy decisions favor proven solutions such as Rhythm360, which offers onboarding in days or weeks instead of the months or years required for internal development. This speed advantage matters because the most common implementation pitfalls, including undocumented verbal consent, ignored 16-day transmission requirements, and untracked opt-out requests, usually arise from rushed manual processes without systematic controls. A mature platform reduces these risks with built-in compliance guardrails that standardize consent and monitoring workflows.
The table below shows how automation addresses these pitfalls at each practice maturity level and helps you decide when manual processes no longer justify the effort.
Maturity Level | Manual Process | Rhythm360 Automation |
Low | Paper consent forms and manual tracking | Digital consent with automatic documentation |
Medium | EHR templates with periodic audits | Real-time compliance monitoring |
High | Dedicated compliance staff | AI-powered, audit-ready automation |
Remote patient monitoring requires documented consent that explains the scope of monitoring, data sharing protocols, cost responsibilities, and opt-out procedures. Teams must obtain this consent before billing CPT 99454 and record it in the medical record with clear details about the monitoring agreement.
The 2026 updates focus on stricter audit-ready documentation while keeping existing consent rules in place. CMS still allows auxiliary personnel to obtain consent under general supervision, and practices must maintain thorough documentation to withstand increased audit scrutiny and avoid claim denials.
CPT 99454 is the monthly RPM code that requires at least 16 days of data transmission in a 30-day period. See the detailed explanation in the “What CPT Code 99454 Covers in Cardiology” section above for device requirements and cardiology-specific applications.
CPT 99454 can be billed once per calendar month per patient when the minimum 16-day data transmission requirement is met. Multiple patients can each generate monthly 99454 claims, but a single patient cannot have more than one 99454 claim in the same calendar month, regardless of monitoring duration.
Clear RPM consent workflows for CPT 99454 protect revenue and support audit-ready claims for cardiology practices. Strong documentation, consistent processes, and automated compliance tools turn consent management from a manual burden into a reliable growth engine.