CPT code 99454 covers device supply with daily recording or alert transmission that meets 16–30 days of data transmission in a 30-day period, with approximately $47 reimbursement for 2026. The code applies to FDA-cleared devices that automatically collect and transmit physiological data and excludes manual patient entries.
Requirement | Details | Cardiology Note |
Data Minimum | At least 16 calendar days in a 30-day period (aggregate transmissions; per CMS 2026 PFS) | FDA devices such as BP cuffs and scales for HF/HTN and integrated CIED systems like CardioMEMS |
Billing Frequency | Once per month per patient, about $47 reimbursement | Bill monthly, even with multiple devices, and align with calendar months |
Device/Consent | FDA-cleared, automatic transmission devices with documented patient consent | Use interactive devices for HF/HTN and document OEM or CIED proof |
2026 Updates | Mutually exclusive with 99445 for 2–15 days, so do not double bill | Rhythm360 auto-logs data for Epic and Cerner workflows |
Bill CPT 99454 when you have at least 16 days of RPM data from FDA-approved devices within a 30-day period. The code requires automatic data transmission, which fits cardiology practices that monitor heart failure patients with weight scales, hypertension patients with blood pressure monitors, and CIED patients with integrated monitoring systems. Rhythm360 captures this data across all major OEMs in a single workflow.
The minimum monitoring duration for CPT 99454 in 2026 remains 16 or more days of data transmission in a 30-day period. This standard ensures enough clinical data for meaningful patient management and supports the monthly reimbursement.
Use this quick compliance checklist:
Cardiology practices that use Rhythm360’s AI-powered platform fill data gaps and reach more than 99.9% compliance for heart failure, hypertension, and CIED monitoring. The system logs transmissions automatically, removes manual tracking, and lowers denial risk. Clear documentation of the 16-day minimum through automated transmission records protects revenue and reduces audit exposure.

Clear distinctions between RPM codes prevent billing errors and support full revenue capture. CPT 99454 covers device supply for 16–30 days, while the other codes address setup and management services.
Code | Description | Frequency | Cardiology Application |
99453 | Initial setup, about $22 | Once per lifetime | Bill with 99454 in the same month when starting RPM |
99454 | Device supply and data, about $47 | Monthly with at least 16 days | RPM devices such as HF scales and BP monitors |
99457 | Twenty or more minutes of management, about $52 | Monthly | Clinician review of CIED and RPM data |
99458 | Each additional 20 minutes, about $41 | Add-on to 99457 | Extended management time |
CPT code 99454 covers device supply and data transmission, while 99457 covers clinical management time. Practices can bill both codes in the same month when they deliver full RPM services. Avoid overlap between CPT 99454 and the new CPT 99445 code, and select the correct code based on actual days of data transmission, either 2–15 days or 16 or more days.
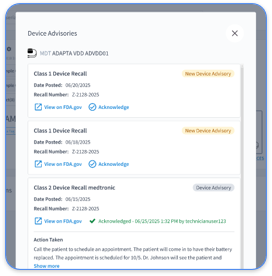
Strong documentation prevents claim denials and audit failures. OIG has increased oversight of RPM billing, so detailed compliance records now matter more than ever.
Use these core documentation elements:
Rhythm360 captures documentation from all major OEMs, connects directly with your EHR, and generates audit-compliant reports. The platform helps prevent the 30% revenue loss that often comes from weak documentation and claim denials.
Schedule a demo to see how automated documentation reduces compliance risk and supports maximum reimbursement.
Cardiology practices often lose significant RPM revenue through preventable billing errors and inefficient workflows. Manual data entry and poor device qualification frequently trigger claim denials.
Watch for these major revenue leak sources:
Practices that rely on manual processes often see about 30% revenue leakage from RPM programs due to administrative overhead, missed transmission days, and documentation gaps. Rhythm360’s unified dashboard removes multi-OEM portal juggling, and AI-powered alerts shorten response times by about 80%, which supports stronger billing compliance and revenue capture.
Rhythm360 delivers vendor-neutral automation that turns fragmented multi-OEM RPM management into a streamlined and profitable program. The platform automatically logs CPT 99454 qualifying events for heart failure, hypertension, and CIED monitoring and supports bi-directional EHR integration and mobile access.
Key advantages:
Case studies show practices capturing up to 300% more RPM revenue through automated documentation, lower administrative overhead, and fewer billing errors. The platform addresses the unique demands of cardiology RPM programs while supporting high compliance and strong profitability.
Schedule a demo today to remove multi-OEM complexity and unlock your practice’s RPM revenue potential.
CPT 99454 requires at least 16 days of data transmission within a 30-day period. For shorter monitoring periods of 2–15 days, use the new CPT 99445 code introduced in 2026. The 16-day requirement provides enough clinical data for meaningful patient management and supports the monthly reimbursement of about $47.
Clinicians can bill CPT 99453 for initial setup and CPT 99454 for device supply and data in the same month. CPT 99453 is a one-time lifetime charge that covers patient education and device setup, while CPT 99454 covers ongoing monthly device supply and data transmission. These codes work together in a complete RPM program.
CMS 2026 guidelines introduce CPT 99445 for 2–15 days of monitoring and keep CPT 99454 for 16 or more days. The codes are mutually exclusive, so bill only one per 30-day period based on actual data transmission days. All RPM devices must be FDA-cleared and support automatic transmission, and manual patient entries do not qualify for reimbursement.
CPT 99454 billing requires device transmission logs that prove at least 16 days of automatic data collection, FDA device identification numbers, patient consent forms, medical necessity documentation, and clinical management records that show how transmitted data influenced care decisions. Maintain detailed audit trails to support compliance and defend revenue.
In cardiology, CPT 99454 covers FDA-approved devices for heart failure monitoring with weight scales and blood pressure monitors, hypertension management, and integration with CIED systems such as CardioMEMS. The code supports continuous cardiovascular monitoring between office visits. Rhythm360’s HF and HTN RPM platform streamlines this work across multiple device manufacturers.
Master CPT 99454 billing with clear checklists and Rhythm360’s automated compliance platform to reach zero denials and up to 300% revenue growth.
Schedule a demo today to strengthen your cardiology RPM profitability and reduce administrative burden.