Last updated: February 24, 2026
Wearable heart monitors fall into four main sensing categories, and each category has distinct accuracy profiles and clinical roles. Photoplethysmography (PPG) devices, including Apple Watch and Fitbit models, measure heart rate with optical sensors that track blood volume changes. PPG wearables like Fitbit Charge 6 reach strong HR accuracy with mean absolute error (MAE) of 4.5 bpm and mean absolute percentage error (MAPE) of 5.5%, which meets clinical accuracy thresholds.
Electrocardiogram (ECG) wearables capture electrical heart activity through skin contact electrodes. Smartwatches with ECG capability and dedicated devices like KardiaMobile fit in this group. Chest strap monitors, such as Polar H10, place ECG sensors across the chest for higher signal quality. Chest strap devices like Hexoskin achieve 87.4% mean HR accuracy over 24 hours, which stays below the 10% threshold for clinical acceptability.
Cardiac patches, including the Zio XT system, deliver continuous ECG monitoring through adhesive electrodes. Zio monitors reach 99% physician agreement on end-of-wear reports and perform strongly for detecting arrhythmias such as atrial fibrillation. Motion artifacts affect every wearable type, although PPG devices show higher error rates during physical activity compared with ECG-based systems.
| Device | Type/FDA Status | Accuracy | Best For |
|---|---|---|---|
| Apple Watch Series 12 | PPG + ECG / FDA-cleared AFib detection | MAPE 6-7% HR, 98% AFib sensitivity | Consumer integration, $399-799 |
| Zio XT Patch | Continuous ECG / FDA Class II | 99% physician agreement | Clinical arrhythmia detection, $300-500 |
| Polar H10 | Chest strap ECG / Non-medical | 87% accuracy, MAPE ~5% | Exercise monitoring, $90 |
| KardiaMobile 12L | Handheld 12-lead ECG / FDA-cleared | 35 FDA-cleared determinations | Point-of-care diagnostics, $149 |
Professional cardiology practices need wearables that combine clinical accuracy with smooth workflow integration. The Zio XT patch remains a leading choice for continuous arrhythmia monitoring, while consumer devices like Apple Watch support broader patient adoption despite limits in clinical-grade accuracy.
Cardiologists primarily choose clinical-grade devices such as Zio patches and Holter monitors for diagnostic use. AI-integrated ECG monitoring platforms introduced by leading manufacturers in 2025 support real-time arrhythmia detection with higher precision in clinical environments. At the same time, practices increasingly fold consumer wearables into RPM workflows as they pursue continuous monitoring beyond traditional episodic assessments.
FDA-authorized wearable sensor-based digital health technology devices for cardiovascular use include Apple’s Irregular Rhythm Notification Feature (IRNF), iRhythm’s ZEUS System (Zio Watch), and AliveCor’s Triangle System. AliveCor received FDA clearance in January 2026 for the Kardia 12L ECG System with 35 FDA-cleared determinations, which expands clinical diagnostic options for handheld ECG devices.
The Zio XT patch gives electrophysiologists continuous ECG monitoring with high sensitivity for arrhythmia detection. Zio’s deep-learned algorithm reaches 99% physician agreement on end-of-wear reports, which supports confident clinical decision-making. The device does not provide real-time alerts, so clinicians review data after the wear period, and this review can delay urgent interventions compared with live-streaming systems.
Clinical wearable ECG monitors deliver stronger signal quality than PPG-based systems, especially when patients move. Single-lead ECG recordings from wearables are noisier than standard 12-lead ECGs and lack full spatial information, which narrows their diagnostic scope. Effective use in practice requires platforms that normalize diverse data formats and convert raw signals into clear, actionable clinical insights.
Wearable heart monitors still present notable limitations in professional cardiology settings. Motion artifacts create high mean absolute error (MAE ≈10–12 years) in AI-ECG age models, which signals non-random error patterns. Continuous skin contact can cause irritation, and short battery life restricts long-term monitoring. Data fragmentation across multiple device platforms creates silos that slow comprehensive patient assessment and reduce workflow efficiency.
False positive alerts increase clinician fatigue and burnout. At the same time, fragmented interoperability between wearable devices and healthcare platforms lowers data usability for clinical decision-making. These gaps highlight the need for vendor-neutral RPM platforms that unify disparate wearable data streams into a single clinical view.
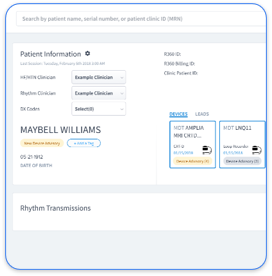
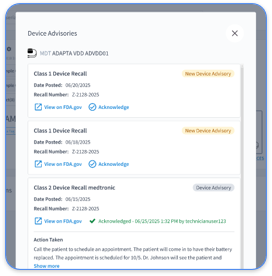
Fragmented data creates operational chaos for cardiology practices that manage multiple device types and manufacturer portals. Rhythm360 solves this problem with vendor-neutral integration that consolidates data from all major CIED manufacturers, including Medtronic, Abbott, and Boston Scientific. The platform reaches more than 99.9% data transmissibility through redundant data feeds, computer vision, and AI-powered extrapolation.
Clinical workflow integration supports real-time alert triage and cuts response times for critical events by up to 80%. When a device detects new-onset atrial fibrillation, Rhythm360’s AI engine prioritizes the alert, generates clinical documentation, and supports same-day anticoagulation initiation. The mobile application gives clinicians secure access to patient data, which helps teams coordinate care from any location.
Implementation uses API connections with existing platforms, EHR synchronization through HL7 protocols, and automated billing documentation for relevant CPT codes. Schedule a demo to see how Rhythm360 converts cardiac device data into practical clinical intelligence.

Updated 2026 RPM service codes include 99453 for initial setup, 99454 for 16-30 days of monitoring, and new Category III code 0962T for AI-assisted cardiac diagnostics via wearable devices. Rhythm360 automates documentation and billing compliance for these codes so practices can capture revenue that previously went unbilled. The platform’s combined approach to wearable integration and automated billing can deliver up to 300% revenue improvement through stronger CPT code capture and lower administrative workload.
Most consumer wearables such as standard fitness trackers do not hold FDA approval for medical diagnosis. Specific features on devices like Apple Watch, including AFib detection, and dedicated medical wearables such as Zio patches have received FDA clearance for defined clinical uses. The key difference lies between general wellness tracking and diagnostic functions that meet FDA standards for medical device classification.
Apple Watch does not replace comprehensive clinical ECG monitoring because it uses a single-lead design and produces more noise than 12-lead clinical systems. The device works well for continuous rhythm tracking and AFib screening. Formal diagnosis still requires the spatial detail and signal quality of traditional ECG equipment. Apple Watch functions as a useful screening tool that prompts clinical confirmation before major decisions.
Rhythm360 connects data from CIEDs and RPM devices through vendor-neutral APIs that standardize diverse data formats from major manufacturers. The platform applies computer vision and AI algorithms to extract clinical insights from different report layouts and merges those findings with other device data in a single clinical dashboard. This approach removes the need for clinicians to log into multiple manufacturer portals.
Cardiologists usually recommend clinical-grade devices such as CIEDs for diagnostic monitoring and RPM programs. The priority involves integrating these varied data sources through platforms like Rhythm360 that deliver clinical-grade data management, alert prioritization, and complete patient monitoring across all supported device types.
Accuracy varies by device type and clinical scenario. Zio patches reach 99% physician agreement for arrhythmia detection. Consumer PPG devices show 84-93% heart rate accuracy and higher error rates during motion. ECG-based wearables generally outperform PPG systems for arrhythmia detection, and all wearable categories benefit from AI analysis and clinical oversight that reduce false positives and improve diagnostic reliability.
The leading wearable heart monitors for cardiology practices in 2026 create strong opportunities for continuous patient monitoring, but their value depends on unified RPM workflows. Devices such as Zio patches, Apple Watch, and Polar monitors each offer distinct strengths, yet fragmented data management limits their impact on outcomes. Rhythm360’s vendor-neutral platform converts scattered cardiac device data into clear clinical intelligence, helping practices reach 80% faster response times and up to 300% revenue improvement. Schedule a demo to see how Rhythm360 can reshape your cardiology practice’s approach to cardiac device integration.